Volume 8, Issue 1 (2022)
Pharm Biomed Res 2022, 8(1): 73-86 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Jumoke Olayemi O, Eloyi John J, Abdullahi R, Yetunde Isimi C. Multifunctional Properties of Co-processed Dioscorea rotundata Starch and Abelmoschus esculentus Fruit Gum in Direct Compression of Metronidazole Tablets. Pharm Biomed Res 2022; 8 (1) :73-86
URL: http://pbr.mazums.ac.ir/article-1-438-en.html
URL: http://pbr.mazums.ac.ir/article-1-438-en.html
1- Department of Pharmaceutical Technology and Raw Materials Development, National Institute for Pharmaceutical Research and Development, Abuja, Nigeria.
Full-Text [PDF 927 kb]
(1081 Downloads)
| Abstract (HTML) (2179 Views)
Full-Text: (1394 Views)
Introduction
Excipients are very important in pharmaceutical formulations. They serve as binders, disintegrants, fillers, glidants, absorbents, and so on to ensure that the prepared formulations are robust, acceptable, and efficient for the intended purpose. However, no single excipient has all the desirable properties required to prepare a stable dosage form. This limitation has led to the development of multifunctional excipients with properties like better flow, more compressibility, low moisture content, and fast disintegrating ability. Because the cost of producing new excipients is high, repurposing or manipulation of already existing excipients has become an exploitable option.
Co-processing is a technique whereby appropriate processes combine two or more proven existing excipients to produce new excipients [1, 2]. The concept of co-processing is based on the interaction of the combined excipients on the sub-particle level without altering the chemical entity of the individual excipients. It brings about materials with improved functionality over the individual excipients and expands their applicability in the pharmaceutical industries. These excipients are Generally Regarded As Safe (GRAS), mainly because the parent excipients are certified safe. Hence, they do not require additional toxicological tests to certify their safety [3, 4].
Excipients produced by co-processing are fast gaining recognition because they can be directly compressed into tablets which is more economical than the tedious wet granulation processing of tablet manufacture. Some commercially available co-processed excipients are as follows. Prosolv® SMCC HD90 is composed of microcrystalline cellulose and colloidal silicon dioxide and causes rapid tablet disintegration when incorporated as a filler-binder in tablet formulations [5]. Ludipress® is composed of lactose monohydrate. Also, Kollidon® 30 and Kollidon® CL are specially developed for direct compression of tablets, while StarCap® is composed of pregelatinized starch and maize starch, which is used in capsule formulations [6, 7].
To develop new, functional, and cost-effective excipients, researchers have exploited the use of natural and readily available materials. Literature reveals several such investigations, including co-processing of neem gum with rice starch and or lactose which produced a free-flowing excipient with enhanced consolidation properties [8]. Co-processing of acacia gum and calcium carbonate produced fast disintegrating tablets [9], while the rapid disintegrating ability of co-processed acacia gum and maize starch in metronidazole tablets has also been reported [10]. In another study, co-processing of starch from Artocarpus altilis fruit with sodium starch glycolate was found to produce an excellent flowing excipient with good direct compressional properties [11]. In a different study [12], mucilage from seeds of Ocimum bascilium co-processed with mannitol produced excipients with excellent superdisintegrating ability in terbutaline sulfate mouth dissolving tablets. Another study [13] revealed that co-processing sweet potato starch with silicon dioxide produced tablets with good mechanical properties while co-processing pregelatinized cocoyam starch and acacia gum showed the potential of producing fast release directly compressed tablets [14]. In another study, Nnabuike ND and Oyeniyi YJ. evaluated the disintegration property of co-processed of Abelmoschus esculentus gum and Manihot esculenta starch in metronidazole tablet formulations prepared by the wet granulation technique [15]. They reported that the co-processed excipient possessed comparable disintegration properties with that of a standard disintegrant. In another study, co-processing some superdisintegrants with moringa gum produced robust and stable orodispersible tablets by direct compression [16]. These studies, among others, show that co-processing native starches with other materials and using appropriate means improve the compact and or specific properties of the resulting excipient.
In the present study, starch from Dioscorea rotundata and gum from Abelmoschus esculentus fruit were used. Starch is a natural and relatively low-cost material obtained from readily available plant sources by simple means. It is widely used in the pharmaceutical industry. However, despite its versatility, native starch is not suitable for direct compression because of its poor compressible properties [8, 12, 17]. Gums, on the other hand, produce tablets with good mechanical strength [18, 19]. Therefore, the combination of both materials is used to obtain an optimum product that could be employed in the direct compression of tablet formulations.
This study aimed to develop co-processed excipients (CYG) by co-fusing starch from Dioscorea rotundata with different concentrations of gum extracted from the pods of the Abelmoschus esculentus fruit. The physical, mechanical, and release properties of the developed excipients were evaluated as multifunctional excipients in directly compressed metronidazole tablet formulations.
Materials and Methods
Study materials
The study materials were metronidazole (BDH Chemicals Ltd Poole, England), Prosolv® (JRS Pharma, Cedar Rapids, Iowa, USA), CombiLac® (Meggle Pharma, Evonik, Wasserburg, Germany), and magnesium stearate (BDH Chemicals Ltd Poole, England). Dioscorea rotundata starch and Abelmoschus esculentus fruit gum were extracted in the National Institute for Pharmaceutical Research and Technology, NIPRD, Abuja, Nigeria. This experimental study was carried out in NIPRD, Abuja, Nigeria, from July to August 2021.
Extraction of Dioscorea rotundata starch
Dioscorea rotundata starch was extracted according to the method previously described [20] with some modifications. The skin of the tubers of Dioscorea rotundata was peeled, washed in water, cut into small pieces, and soaked in sodium metabisulfite solution (0.75 %w/v) at a ratio of 1:2 (yam pieces: sodium metabisulfite solution) overnight. The soaked yam pieces were wet-milled. Then, the milled mixture was sieved with a muslin cloth, and the resulting suspension was centrifuged at 1500 rpm for 15 min (Heraeus Sepatech Labofuge Ae, Germany). The supernatant was poured out, the sediment starch coded as WYS was air-dried at room temperature for 12 h and then dried in the oven at 400C for 2 h. The dried starch was pulverized, packaged into air-tight containers, and stored in a desiccator until further use.
Extraction of Abelmoschus esculentus fruit gum
Abelmoschus esculentus fruit gum was extracted using the method described by an earlier study [21]. Fresh fruits of Abelmoschus esculentus were washed, their seeds were removed, and the cut pods were soaked in distilled water for 12 h. Afterward, the mixture was filtered using a muslin cloth; the pod residues were discarded while the filtered viscous extract was precipitated with acetone in a ratio of 1:3 (mucilage to acetone). The precipitation process using acetone was repeated three times to ensure the resulting mass (Abelmoschus esculentus gum) was devoid of water. The gum coded as OKG was air-dried for 12 h, pulverized, packaged into air-tight containers, and stored in a desiccator until further use.
Co-processing of Dioscorea rotundata starch and Abelmoschus esculentus fruit gum
Tablet batches of 100 g containing Dioscorea rotundata starch and Abelmoschus esculentus fruit gum at different ratios (99:1, 97:3, and 95:5) were prepared using a combination of two methods [17, 22] with some modifications. The appropriate weight of WYS (99 g) was dispersed in distilled water to obtain 40% w/v starch dispersion and heated at 70°C for 20 min to get a paste. A dispersion of OKG (1 g) made in distilled water (50 mL) at room temperature was added to the WYS dispersion at 40°C and stirred until a thick mass was obtained (20 min). The resulting mass (CYG1) was air-dried for 24 h, then dried in the oven at 40°C for 2 h, pulverized, passed through a sieve (250 μm), and kept in the desiccator until further use. Other batches of co-processed excipients (CYG3 and CYG5) were prepared using the appropriate quantities of WYG and OKG.
Evaluation of co-processed excipients
Fourier Transform Infra-Red (FTIR) spectra studies
The co-processed excipient (CYG1), metronidazole, and combination of CYG1 and metronidazole were triturated with potassium bromide powder made into pellets (1 ton/cm2). Infra-Red (IR) spectra were obtained between scanning ranges of 4000 and 400 cm-1 from the Magna-IR, 560 spectrometers (Perkin Elmer, USA).
Flow properties of the co-processed excipients
Angle of repose
Ten grams of each co-processed (CYG) batch was poured into a funnel whose orifice had been plugged; the height and diameter of the heap formed after opening the orifice were determined, and the angle of repose (A) was calculated from the following equation (Equation 1);
.jpg)
Bulk and tapped densities
The volume occupied by 10 g from each batch of the excipient in a measuring cylinder (100 mL) was recorded as the bulk volume. The measuring cylinder was tapped 100 times using the Stampfvolumeter (STAV 2003JEF, Germany), and the volume after tapping was recorded as the tapped volume. Bulk Density (BD) and Tapped Density (TD) were calculated as shown below (Equations 2, 3):
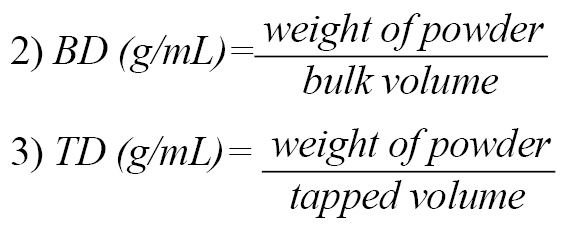
Compressibility Index (CI) and Hausner Ratio (HR)
These parameters were calculated from data obtained from the bulk and tapped densities (Equations 4, 5):

True Density
The liquid displacement method was adopted as described by Olayemi et al. [23]. Liquid paraffin was employed as the displacement fluid. Three determinations were made for each batch of excipient, and True Density (Trd) was computed as shown below (Equation 6):
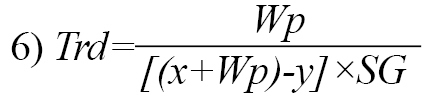
, where Wp is the weight of starch powder, x is the weight of the bottle and fluid, y refers to the weight of the bottle, fluid and starch powder, and SG is the specific gravity of liquid paraffin (0.865 g/mL).
Swelling Capacity (SC)
This capacity was determined using an earlier method [24]. The tapped volume occupied by 5 g of each batch of excipient placed in a measuring cylinder (100 mL) was noted. The powder was dispersed in water (85 mL), then made up to volume and allowed to stand at room temperature for 24 h. Then, the volume of the sediment was noted, and swelling capacity was computed using the equation below (Equation 7):

Hydration Capacity
One gram of each batch of excipient was placed in a centrifuging tube, and distilled water (10 mL) was poured into the tube. The tube was covered, shaken intermittently for 2 min, and centrifuged at 3000 rpm for 10 min. The supernatant was decanted, and the weight of the sediment (wet starch) was determined. Hydration capacity was the ratio of the wet starch weight to that of the dry sample.
Preparation of Metronidazole tablets
The excipients were incorporated into multifunctional (filler, binder, disintegrant) in metronidazole tablet formulations prepared by direct compression. Thirty tablets with a target weight of 500 mg each were produced for each of the batches of excipient. The composition of ingredients for tablet preparation is listed in Table 1.
.jpg)
The appropriate quantity of metronidazole powder was weighed into a porcelain mortar and mixed with CYG1 powder in geometric measures until a homogenous powder mix was obtained. The lubricant (magnesium stearate) was added to the powdered, mixed, and then compacted into tablets at 8 Nm-2 in the Manesty tableting machine (Shanghai, China) using the 10-mm Punch and Die set. The same procedure was undertaken to prepare tablets of the other batches containing CYG3 and CYG5. The produced tablets were kept for 24 h before evaluation to allow elastic recovery.
Evaluation of Metronidazole tablets
Uniformity of weight
Ten tablets were randomly selected from each batch and weighed; the average weight and standard deviation from the mean were determined.
Crushing strength
The crushing strength/hardness (N) of five randomly selected tablets from each batch was determined using the Monsanto hardness tester, then the mean was calculated.
Friability test
Five tablets from a batch were collectively weighed (W1), placed into the Erweka Friabilator, and allowed to rotate at 25 rpm for 4 min. Afterward, the tablets were de-dusted, re-weighed (W2), and friability (%) was calculated as follows (Equation 8):
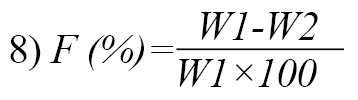
The same procedure was performed for the other batches of tablets.
Crushing strength-friability
This parameter was computed as the product of the tablet crushing strength and friability.
Disintegration test
One tablet from each batch was placed in each of the six compartments of the disintegration tester (Erweka ZT4-4, Germany) containing distilled water thermostated at 37±0.5ºC as the medium. The time taken for all tablet particles to pass through the compartment’s mesh was noted, and the average was determined as the disintegration time. The same procedure was performed for the other batches of tablets.
Determination of Disintegration Efficiency Ratio (DER)
The Disintegration Efficiency Ratio (DER) was determined using the relationship (Equation 9):
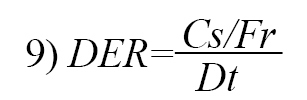
While the dimensionless disintegrant quantity (DERc) was calculated using the equation below (Equation 10):
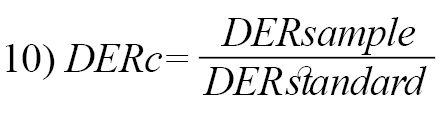
, where Cs refers to crushing strength, Fr is friability, and Dt refers to disintegration time.
In Vitro dissolution studies
Standard concentrations of metronidazole in 0.1 N HCl were made between 0.5 µg/mL and 60 µg/mL. The corresponding absorbance was obtained from the UV-visible spectrophotometer (Cary 60) at a λmax of 230 nm.
One tablet from each batch was placed in the dissolution basket and lowered into the dissolution vessel containing 0.1 N HCl (900 mL) maintained at 37±0.5°C. The apparatus (Dissolution tester, RC-6, India) was set to rotate at 100 rpm; aliquots of 5 mL were withdrawn at intervals of 5, 10, 15, 30, 45, and 60 min and replaced with an equal volume of the medium. The withdrawn samples were filtered through a 0.4-µm membrane filter, and the absorbance was determined at 230 nm using the UV-visible spectrophotometer (Cary 60). The concentration of metronidazole was determined using the calibration curve.
Statistical analysis
Data obtained from the study were expressed as Mean±SD. Statistical considerations for the dissolution profiles were analyzed using the similarity fit factors [25] and calculated using the equation below (Equation 11):
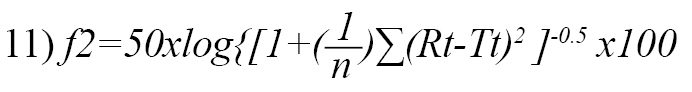
, where Rt refers to the cumulative percentage of reference product dissolved at time t, Tt is the cumulative percentage of test product dissolved at time t, and n denotes the number of time points.
Results
Flow properties of the prepared excipients
Table 2 presents the flow and swelling properties of Dioscorea rotundata starch (WYS), Abelmoschus esculentus gum (OKG), and the co-processed excipients (CYG1, CYG3, and CYG5).
.jpg)
The angle of WYS repose (24.39°) was higher than that of OKG (22.93°), while those of CYG1, CYG3, and CYG5 were 19.04°, 21.95°, and 24.29o, respectively. However, all values fell within the limit for excellent flow. Carr’s compressibility indexes for CYG1, CYG3, and CYG5 were between 11.66% and 19.58%, which showed they are not cohesive and are readily compressible. This result was further expressed with Hausner ratio values between 1.13 and 1.24.
Hydration and swelling capacity of the prepared excipients
According to Table 2, the co-processed excipients had appreciably higher hydration capacity (2.53-2.74) than WYS (1.19), although OKG showed the highest hydration capacity (11.19). Correspondingly, assessment of the ability of the excipients to swell was found to be appreciably higher in the co-processed excipients (170% - 200%) than WYS (80%), while OKG also exhibited the most elevated swelling (250%).
FTIR
FTIR spectra of metronidazole (Figure 1A) show peaks at 3205.5 cm-1, 3101.1 cm-1, and 1535.7 cm-1, with other peaks at 1472.3 cm-1, 1423.3 cm-1, and 1367.9 cm-1 representing several molecular associations. Spectra of the co-processed excipient presented in Figure 1B show characteristic absorption vibrations between 741.7 cm-1 and 991.5 cm-1, representing the finger-print region of the excipient. Broad bands at 350 cm-1 and 3000 cm-1 and peaks at 2929.7 cm-1 and 1636.7 cm-1 were also observed.
.jpg)
Properties of Metronidazole tablets
The average weights of the tablets were between 490.60 and 494.90 mg (Table 3), which is within the official limit for tablets of 500 mg target weight.
.jpg)
Tablet hardness for all batches ranged between 38.44 and 73.26 N, although MTP prepared with Prosolv© was the hardest (73.26 N) of them. Increasing the amount of OKG in the co-processed excipients increased the hardness of the tablets (38.55<55.90=55.90 N) as observed for MT1, MT3, and MT5, respectively.
Tablet friability was between 0.82% and 1.54% for all the batches (Table 3). Those tablets prepared with the co-processed excipients (MT1, MT3, and MT5) were more friable (1.50%, 1.33%, and 1.41%, respectively) than MTC and MTP (0.82% and 0.94%, respectively). However, the Crushing Strength-Friability Ratio (CSFR) showed MT3 to be the strongest tablet with a value of 42.03, followed by 39.65 for MT5 and 25.63 for MT1. Values of MTC and MTP, on the other hand, had CSFR values of 31.20 and 77.93, respectively.
Tablet disintegration was between 33 and 111 s, with MT1, MT3, and MT5 disintegrating at 33.33, 33.83, and 63.87 s, respectively (Table 3). This condition portrays the ability of the co-processed excipients to elicit faster disintegration than Prosolv® (111.50 s). On the other hand, MT1 and MT3 had similar disintegration times (33.33 and 33.67 s, respectively) with MTC (33.67 s). Disintegration Efficiency Ratio (DER) values of MT1 and MT3 are 1.19 and 1.78, respectively, greater than the value 1 set as a lower limit for better disintegrating excipient. However, MT5 had a value of 0.89.
The fraction of drug released after 30 min, DE30 min, was lower in tablets containing the co-processed excipients than those containing the reference excipients (Table 3). However, MT1 had similar efficiency to MTC and MTP. At the end of 60 min dissolution, however, drug release from all the tablets was between 87.90% and 99.38% (Figure 2).
.jpg)
The rank order for drug release was MT1>MT5>MTP>MT3>MTC showing drug release from MT1 was higher than from similar tablet formulations (MT3 and MT5) or tablets prepared with reference excipients (MTC and MTP).
According to Table 4, f2 values are between 39.39% and 48.92% when the dissolution profile of tablets prepared with the co-processed (MT1, MT3, and MT5) was compared with those prepared with CombiLac© (MTC).
.jpg)
However, compared to tablets prepared with Prosolv® (MTP), the values fell between 47.22% and 65.67%.
Table 5 presents that the release kinetics for MT1, MT3, MT5, and MTP favors the Higuchi kinetics while that of MTC favors the first-order kinetics. However, the mechanism of drug release as determined by the Korsmeyer-Peppas model shows “n” values between 0.5836 and 0.6796, indicating that drug release was by Non-Fickian diffusion.
.jpg)
Discussion
The angle of repose reflects the ability of a powder to flow. It is measured by powder resistance to flow. Limits set for this measurement indicate that materials with angles ≤300 have excellent flow, those with values between 31º and 35o good flow, those between 36º and 40º fair flow, and those with values of ≥400 poor flow [26]. The presence of OKG in the co-processed excipients is observed to improve their flow.
Carr’s compressibility index indicates the propensity of powdered materials to decrease under pressure. The materials with values above 25% are cohesive and have poor flow. The results show that CYG excipients can be easily compressed under pressure with values between 11.66% and 19.58%. Similarly, the excipients were non-cohesive and possessed fair flow, as expressed by the Hausner ratio between 1.13 and 1.24 [27].
Okra gum (OKG) had higher hydration capacity and swelling ability attributable to the fact that gums like OKG have charged hydrogen atoms which attract water molecules resulting in marked swelling [28]. Increasing the concentration of OKG in the co-processed batches resulted in the increased swelling ability due to particle agglomeration and mechanical interlocking of the particles. This observation is in line with that of a recent study [29], where the addition of Okra gum to pregelatinized potato starch enhanced swelling of the co-processed product. Swelling is not only one mechanism of disintegration but is also a precursor to prolonged tablet disintegration due to the formation of a gelatinous layer around the tablets when it comes in contact with an aqueous medium [30].
FTIR spectra of metronidazole (Figure 1A) show characteristic peaks at 3205.5, indicating O-H stretching, at 3101.1 C=H stretch, while at 1535.7, the C=N vibration. The peaks representing 1472.3, 1423.3, and 1367.9 represent C=C aromatic ring vibrations, CH3 bending vibrations, and C-N stretch vibrations. In contrast, those around 1185.3, 1073.5, and 823.7 represent C-O stretch, C-O bond, and C-H aromatic vibrations which are consistent for metronidazole [31]. The spectra obtained for the co-processed excipient in Figure 1B are characteristic of the co-processed excipient and can identify the excipient. The spectra representing the combination of the co-processed excipient (Figure 1C) and metronidazole show some of the characteristic bonds of metronidazole. However, no new peaks were observed, indicating no interaction between the co-processed excipient and metronidazole.
The average weights of the tablets listed in Table 3 show none of the formulated tablets deviated from the average weight by 5% according to official specification [32]. Thus, the tablets have uniform weights. This finding shows that combining the co-processed excipients and metronidazole can be compressed under pressure without varying tablet weights.
Tablet hardness/crushing strength is a parameter that symbolizes the mechanical strength of the tablet. It reflects the property of a tablet to withstand transportation, storage, and handling [33]. An acceptable hardness for uncoated immediate release tablets is approximately 44.15 N [34]. However, this value could differ depending on the type of excipients used in the formulation. Hardness is associated with disintegration. If a tablet is too hard, it will not disintegrate or require a longer time to decompose. Soft tablets would disintegrate faster and would not withstand the stress of packaging or handling [35]. The results in Table 3 show that increasing the amount of OKG in CYG excipients increases the hardness of the tablets in the formulation in the order of MT1 Friability measures the tendency of tablets to undergo abrasion, stress, and fragmentation [37], which could affect the appearance and acceptability of the tablets. The results show that MT1, MT3, and MT5 were friable, having values greater than the official limit of ≤1% [38]. However, the Crushing Strength-Friability Ratio (CSFR) is a better index in assessing the mechanical strength; tablets with the highest CSFR values represent the strongest tablets [39]. Using this parameter, MT1 is the weakest of them, while MT3 and MT5 have better mechanical strength than MTC. A higher concentration of the gum in the co-processed excipients has created more solid bonds leading to stronger tablets. However, MTP proved to be the strongest. This result suggests that the CYG excipients tend to formulate tablets with good mechanical strength.
Disintegration is the mechanical break-up of a tablet into particles upon contact with an aqueous medium. It is often associated with dissolution, where the drug is available for therapeutic action. Increasing the amount of OKG in the excipients prolongs the time taken for disintegration. The longer disintegration times observed with MT3 could be ascribed to the high mechanical strength of the batch because the stronger the tablets, the more difficult it becomes for the disintegration fluid to penetrate the tablets resulting in prolonged disintegration time.
Incorporation of the CYG excipients in the tablet formulations resulted in faster disintegration than those tablets prepared with a commercial superdisintegrant, Prosolv®. The case of the latter may be attributed to the low disintegration efficiency of microcrystalline cellulose and high tablet hardness, as observed in this study and also reported in other studies [40]. Disintegration times of MT1 and MT3 were similar to those of the co-processed multifunctional excipient CombiLac©. This result portrays the capability of the co-processed excipient (CYG) in effecting fast tablet disintegration, which is comparable with a previous study where prepared cross-linked starch disintegrates faster than Prosolv® [41] and that of Nnabuike and Oyeniyi [15] where Okra gum and cassava starch were co-processed. However, the finding was different from another study where similarly prepared co-processed excipients did not elicit tablet disintegration at the required official time [42].
Similar to crushing strength, the Disintegration Efficiency Ratio (DER) is an acceptable measure of the balance between the tablet’s strength and the disintegration property [43]. Values greater than 1 indicate that the investigated disintegrant has better property than the reference disintegrant. This effect shows that the co-processed excipients CYG1 and CYG3 have better disintegration properties than Prosolv®. In addition, the results show that CYG excipients can be used as a filler to bulk up tablet formulations; it possesses good binding ability by producing powerful tablets and fast tablet disintegrating ability.
Figure 2 shows the dissolution profile of the prepared metronidazole tablets. Dissolution efficiency at 30 min shows slower drug release from MT1, MT3, and MT5 than from MTC and MTP. However, at the end of the dissolution period, the rank order of drug release was MT1>MT5>MT3. Drug release from MT1 was higher than those of MT3 and MT5 and comparable to that of MTC but different from that of MTP.
Similarity fit factor (f2) is used to compare dissolution profiles of a test and reference formulation to indicate sameness or equivalence between both formulations. Values of f2 less than 50% suggest that the dissolution profiles of the compared formulations are different; values equal to 100 % depict identical profiles while those between 50% and 100% show similar profiles [25]. Table 4 presents all the values that were less than 50%, indicating differences in the dissolution profiles of the tablets prepared with the co-processed excipients (MT1, MT3, and MT5) and those prepared with CombiLac® (MTC). These differences could be attributed to the faster dissolution of MTC than the former formulations. On the other hand, only the profiles of MT3 and MT5 were similar to that of MTP prepared with Prosolv®; the dissolution profile of MT1 prepared with CYG1 varied from that of MTP as displayed in Figure 2.
The in vitro release data were fitted into the zero-order, first-order, Higuchi, and Korsmeyer-Peppas models. Then, the model with the highest coefficient was taken as the appropriate model to describe the possible mechanism of drug release. The results show Higuchi model had the highest co-efficient for MT1, MT3, MT5, and MTP. This finding implies a higher drug concentration in the tablet matrix than in the surrounding solution, even though drug diffusion is proposed to have occurred at a constant rate [44]. This result can explain why the drug release was slower at the initial stages of the dissolution studies for MT1, MT3, and MT5 than MTC. On the other hand, the highest co-efficient for MTC was the first order, where the rate of release was dependent on drug concentration within the tablet and could be the reason for the decline in drug release observed with MTC between 20 and 60 min. The release mechanism shows the exponent “n” values to be between 0.49 and 0.89, which indicates non-Fickian diffusion [41]. It suggests that drug release from the prepared tablets was controlled by swelling and diffusion mechanisms.
At the end of dissolution, CYG1 showed the fastest drug release, indicating a relationship between the disintegration of tablets containing CYG1 and dissolution. Quick disintegration increased the tablet dissolution rate by rapidly exposing the particles to the dissolution medium. In this study, drug release from all tablet formulations was within official specification [38] since the amount of metronidazole released was greater than 85%.
Conclusion
Co-processing Dioscorea rotundata starch and Abelmoschus esculentus fruit gum produced a multifunctional directly compressible excipient which could make robust tablets with superior disintegrant property to Prosolv®, a commercial superdisintegrant and comparable properties with CombiLac©, and a novel multifunctional excipient. This study shows the suitability of using cheap and readily available materials to produce a multifunctional excipient for direct compression of oral tablet formulations.
Ethical Considerations
Compliance with ethical guidelines
There were no ethical considerations to be considered in this research.
Funding
No external funding was received for this study. The authors provided the resources used for the study.
Authors' contributions
Conceptualization and study design: Olubunmi J. Olayemi; Investigation, data collection, and analysis: Judith E. John and Rashidat Abdullahi; Supervision and writing the original draft: Olubunmi J. Olayemi and Christianah Y. Isimi; Reviewing and final writing: Olubunmi J. Olayemi, Judith E. John, Rashidat Abdullahi, and Christianah Y. Isimi.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors thank the National Institute for Pharmaceutical Research and Development (NIPRD), Abuja, Nigeria, for providing the facilities to conduct this study.
References
Excipients are very important in pharmaceutical formulations. They serve as binders, disintegrants, fillers, glidants, absorbents, and so on to ensure that the prepared formulations are robust, acceptable, and efficient for the intended purpose. However, no single excipient has all the desirable properties required to prepare a stable dosage form. This limitation has led to the development of multifunctional excipients with properties like better flow, more compressibility, low moisture content, and fast disintegrating ability. Because the cost of producing new excipients is high, repurposing or manipulation of already existing excipients has become an exploitable option.
Co-processing is a technique whereby appropriate processes combine two or more proven existing excipients to produce new excipients [1, 2]. The concept of co-processing is based on the interaction of the combined excipients on the sub-particle level without altering the chemical entity of the individual excipients. It brings about materials with improved functionality over the individual excipients and expands their applicability in the pharmaceutical industries. These excipients are Generally Regarded As Safe (GRAS), mainly because the parent excipients are certified safe. Hence, they do not require additional toxicological tests to certify their safety [3, 4].
Excipients produced by co-processing are fast gaining recognition because they can be directly compressed into tablets which is more economical than the tedious wet granulation processing of tablet manufacture. Some commercially available co-processed excipients are as follows. Prosolv® SMCC HD90 is composed of microcrystalline cellulose and colloidal silicon dioxide and causes rapid tablet disintegration when incorporated as a filler-binder in tablet formulations [5]. Ludipress® is composed of lactose monohydrate. Also, Kollidon® 30 and Kollidon® CL are specially developed for direct compression of tablets, while StarCap® is composed of pregelatinized starch and maize starch, which is used in capsule formulations [6, 7].
To develop new, functional, and cost-effective excipients, researchers have exploited the use of natural and readily available materials. Literature reveals several such investigations, including co-processing of neem gum with rice starch and or lactose which produced a free-flowing excipient with enhanced consolidation properties [8]. Co-processing of acacia gum and calcium carbonate produced fast disintegrating tablets [9], while the rapid disintegrating ability of co-processed acacia gum and maize starch in metronidazole tablets has also been reported [10]. In another study, co-processing of starch from Artocarpus altilis fruit with sodium starch glycolate was found to produce an excellent flowing excipient with good direct compressional properties [11]. In a different study [12], mucilage from seeds of Ocimum bascilium co-processed with mannitol produced excipients with excellent superdisintegrating ability in terbutaline sulfate mouth dissolving tablets. Another study [13] revealed that co-processing sweet potato starch with silicon dioxide produced tablets with good mechanical properties while co-processing pregelatinized cocoyam starch and acacia gum showed the potential of producing fast release directly compressed tablets [14]. In another study, Nnabuike ND and Oyeniyi YJ. evaluated the disintegration property of co-processed of Abelmoschus esculentus gum and Manihot esculenta starch in metronidazole tablet formulations prepared by the wet granulation technique [15]. They reported that the co-processed excipient possessed comparable disintegration properties with that of a standard disintegrant. In another study, co-processing some superdisintegrants with moringa gum produced robust and stable orodispersible tablets by direct compression [16]. These studies, among others, show that co-processing native starches with other materials and using appropriate means improve the compact and or specific properties of the resulting excipient.
In the present study, starch from Dioscorea rotundata and gum from Abelmoschus esculentus fruit were used. Starch is a natural and relatively low-cost material obtained from readily available plant sources by simple means. It is widely used in the pharmaceutical industry. However, despite its versatility, native starch is not suitable for direct compression because of its poor compressible properties [8, 12, 17]. Gums, on the other hand, produce tablets with good mechanical strength [18, 19]. Therefore, the combination of both materials is used to obtain an optimum product that could be employed in the direct compression of tablet formulations.
This study aimed to develop co-processed excipients (CYG) by co-fusing starch from Dioscorea rotundata with different concentrations of gum extracted from the pods of the Abelmoschus esculentus fruit. The physical, mechanical, and release properties of the developed excipients were evaluated as multifunctional excipients in directly compressed metronidazole tablet formulations.
Materials and Methods
Study materials
The study materials were metronidazole (BDH Chemicals Ltd Poole, England), Prosolv® (JRS Pharma, Cedar Rapids, Iowa, USA), CombiLac® (Meggle Pharma, Evonik, Wasserburg, Germany), and magnesium stearate (BDH Chemicals Ltd Poole, England). Dioscorea rotundata starch and Abelmoschus esculentus fruit gum were extracted in the National Institute for Pharmaceutical Research and Technology, NIPRD, Abuja, Nigeria. This experimental study was carried out in NIPRD, Abuja, Nigeria, from July to August 2021.
Extraction of Dioscorea rotundata starch
Dioscorea rotundata starch was extracted according to the method previously described [20] with some modifications. The skin of the tubers of Dioscorea rotundata was peeled, washed in water, cut into small pieces, and soaked in sodium metabisulfite solution (0.75 %w/v) at a ratio of 1:2 (yam pieces: sodium metabisulfite solution) overnight. The soaked yam pieces were wet-milled. Then, the milled mixture was sieved with a muslin cloth, and the resulting suspension was centrifuged at 1500 rpm for 15 min (Heraeus Sepatech Labofuge Ae, Germany). The supernatant was poured out, the sediment starch coded as WYS was air-dried at room temperature for 12 h and then dried in the oven at 400C for 2 h. The dried starch was pulverized, packaged into air-tight containers, and stored in a desiccator until further use.
Extraction of Abelmoschus esculentus fruit gum
Abelmoschus esculentus fruit gum was extracted using the method described by an earlier study [21]. Fresh fruits of Abelmoschus esculentus were washed, their seeds were removed, and the cut pods were soaked in distilled water for 12 h. Afterward, the mixture was filtered using a muslin cloth; the pod residues were discarded while the filtered viscous extract was precipitated with acetone in a ratio of 1:3 (mucilage to acetone). The precipitation process using acetone was repeated three times to ensure the resulting mass (Abelmoschus esculentus gum) was devoid of water. The gum coded as OKG was air-dried for 12 h, pulverized, packaged into air-tight containers, and stored in a desiccator until further use.
Co-processing of Dioscorea rotundata starch and Abelmoschus esculentus fruit gum
Tablet batches of 100 g containing Dioscorea rotundata starch and Abelmoschus esculentus fruit gum at different ratios (99:1, 97:3, and 95:5) were prepared using a combination of two methods [17, 22] with some modifications. The appropriate weight of WYS (99 g) was dispersed in distilled water to obtain 40% w/v starch dispersion and heated at 70°C for 20 min to get a paste. A dispersion of OKG (1 g) made in distilled water (50 mL) at room temperature was added to the WYS dispersion at 40°C and stirred until a thick mass was obtained (20 min). The resulting mass (CYG1) was air-dried for 24 h, then dried in the oven at 40°C for 2 h, pulverized, passed through a sieve (250 μm), and kept in the desiccator until further use. Other batches of co-processed excipients (CYG3 and CYG5) were prepared using the appropriate quantities of WYG and OKG.
Evaluation of co-processed excipients
Fourier Transform Infra-Red (FTIR) spectra studies
The co-processed excipient (CYG1), metronidazole, and combination of CYG1 and metronidazole were triturated with potassium bromide powder made into pellets (1 ton/cm2). Infra-Red (IR) spectra were obtained between scanning ranges of 4000 and 400 cm-1 from the Magna-IR, 560 spectrometers (Perkin Elmer, USA).
Flow properties of the co-processed excipients
Angle of repose
Ten grams of each co-processed (CYG) batch was poured into a funnel whose orifice had been plugged; the height and diameter of the heap formed after opening the orifice were determined, and the angle of repose (A) was calculated from the following equation (Equation 1);
.jpg)
Bulk and tapped densities
The volume occupied by 10 g from each batch of the excipient in a measuring cylinder (100 mL) was recorded as the bulk volume. The measuring cylinder was tapped 100 times using the Stampfvolumeter (STAV 2003JEF, Germany), and the volume after tapping was recorded as the tapped volume. Bulk Density (BD) and Tapped Density (TD) were calculated as shown below (Equations 2, 3):
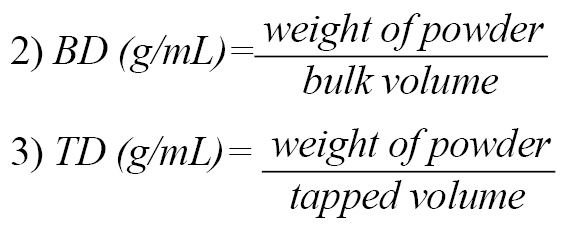
Compressibility Index (CI) and Hausner Ratio (HR)
These parameters were calculated from data obtained from the bulk and tapped densities (Equations 4, 5):

True Density
The liquid displacement method was adopted as described by Olayemi et al. [23]. Liquid paraffin was employed as the displacement fluid. Three determinations were made for each batch of excipient, and True Density (Trd) was computed as shown below (Equation 6):
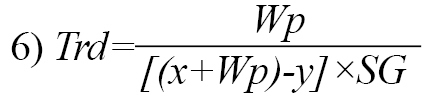
, where Wp is the weight of starch powder, x is the weight of the bottle and fluid, y refers to the weight of the bottle, fluid and starch powder, and SG is the specific gravity of liquid paraffin (0.865 g/mL).
Swelling Capacity (SC)
This capacity was determined using an earlier method [24]. The tapped volume occupied by 5 g of each batch of excipient placed in a measuring cylinder (100 mL) was noted. The powder was dispersed in water (85 mL), then made up to volume and allowed to stand at room temperature for 24 h. Then, the volume of the sediment was noted, and swelling capacity was computed using the equation below (Equation 7):

Hydration Capacity
One gram of each batch of excipient was placed in a centrifuging tube, and distilled water (10 mL) was poured into the tube. The tube was covered, shaken intermittently for 2 min, and centrifuged at 3000 rpm for 10 min. The supernatant was decanted, and the weight of the sediment (wet starch) was determined. Hydration capacity was the ratio of the wet starch weight to that of the dry sample.
Preparation of Metronidazole tablets
The excipients were incorporated into multifunctional (filler, binder, disintegrant) in metronidazole tablet formulations prepared by direct compression. Thirty tablets with a target weight of 500 mg each were produced for each of the batches of excipient. The composition of ingredients for tablet preparation is listed in Table 1.
.jpg)
The appropriate quantity of metronidazole powder was weighed into a porcelain mortar and mixed with CYG1 powder in geometric measures until a homogenous powder mix was obtained. The lubricant (magnesium stearate) was added to the powdered, mixed, and then compacted into tablets at 8 Nm-2 in the Manesty tableting machine (Shanghai, China) using the 10-mm Punch and Die set. The same procedure was undertaken to prepare tablets of the other batches containing CYG3 and CYG5. The produced tablets were kept for 24 h before evaluation to allow elastic recovery.
Evaluation of Metronidazole tablets
Uniformity of weight
Ten tablets were randomly selected from each batch and weighed; the average weight and standard deviation from the mean were determined.
Crushing strength
The crushing strength/hardness (N) of five randomly selected tablets from each batch was determined using the Monsanto hardness tester, then the mean was calculated.
Friability test
Five tablets from a batch were collectively weighed (W1), placed into the Erweka Friabilator, and allowed to rotate at 25 rpm for 4 min. Afterward, the tablets were de-dusted, re-weighed (W2), and friability (%) was calculated as follows (Equation 8):
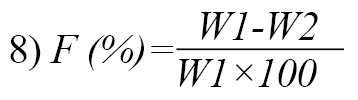
The same procedure was performed for the other batches of tablets.
Crushing strength-friability
This parameter was computed as the product of the tablet crushing strength and friability.
Disintegration test
One tablet from each batch was placed in each of the six compartments of the disintegration tester (Erweka ZT4-4, Germany) containing distilled water thermostated at 37±0.5ºC as the medium. The time taken for all tablet particles to pass through the compartment’s mesh was noted, and the average was determined as the disintegration time. The same procedure was performed for the other batches of tablets.
Determination of Disintegration Efficiency Ratio (DER)
The Disintegration Efficiency Ratio (DER) was determined using the relationship (Equation 9):
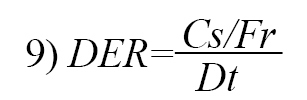
While the dimensionless disintegrant quantity (DERc) was calculated using the equation below (Equation 10):
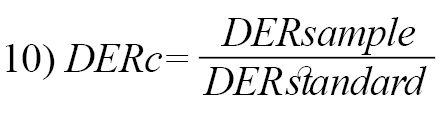
, where Cs refers to crushing strength, Fr is friability, and Dt refers to disintegration time.
In Vitro dissolution studies
Standard concentrations of metronidazole in 0.1 N HCl were made between 0.5 µg/mL and 60 µg/mL. The corresponding absorbance was obtained from the UV-visible spectrophotometer (Cary 60) at a λmax of 230 nm.
One tablet from each batch was placed in the dissolution basket and lowered into the dissolution vessel containing 0.1 N HCl (900 mL) maintained at 37±0.5°C. The apparatus (Dissolution tester, RC-6, India) was set to rotate at 100 rpm; aliquots of 5 mL were withdrawn at intervals of 5, 10, 15, 30, 45, and 60 min and replaced with an equal volume of the medium. The withdrawn samples were filtered through a 0.4-µm membrane filter, and the absorbance was determined at 230 nm using the UV-visible spectrophotometer (Cary 60). The concentration of metronidazole was determined using the calibration curve.
Statistical analysis
Data obtained from the study were expressed as Mean±SD. Statistical considerations for the dissolution profiles were analyzed using the similarity fit factors [25] and calculated using the equation below (Equation 11):
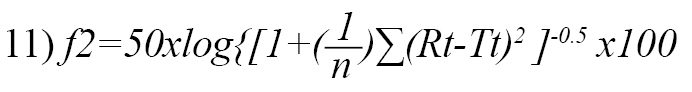
, where Rt refers to the cumulative percentage of reference product dissolved at time t, Tt is the cumulative percentage of test product dissolved at time t, and n denotes the number of time points.
Results
Flow properties of the prepared excipients
Table 2 presents the flow and swelling properties of Dioscorea rotundata starch (WYS), Abelmoschus esculentus gum (OKG), and the co-processed excipients (CYG1, CYG3, and CYG5).
.jpg)
The angle of WYS repose (24.39°) was higher than that of OKG (22.93°), while those of CYG1, CYG3, and CYG5 were 19.04°, 21.95°, and 24.29o, respectively. However, all values fell within the limit for excellent flow. Carr’s compressibility indexes for CYG1, CYG3, and CYG5 were between 11.66% and 19.58%, which showed they are not cohesive and are readily compressible. This result was further expressed with Hausner ratio values between 1.13 and 1.24.
Hydration and swelling capacity of the prepared excipients
According to Table 2, the co-processed excipients had appreciably higher hydration capacity (2.53-2.74) than WYS (1.19), although OKG showed the highest hydration capacity (11.19). Correspondingly, assessment of the ability of the excipients to swell was found to be appreciably higher in the co-processed excipients (170% - 200%) than WYS (80%), while OKG also exhibited the most elevated swelling (250%).
FTIR
FTIR spectra of metronidazole (Figure 1A) show peaks at 3205.5 cm-1, 3101.1 cm-1, and 1535.7 cm-1, with other peaks at 1472.3 cm-1, 1423.3 cm-1, and 1367.9 cm-1 representing several molecular associations. Spectra of the co-processed excipient presented in Figure 1B show characteristic absorption vibrations between 741.7 cm-1 and 991.5 cm-1, representing the finger-print region of the excipient. Broad bands at 350 cm-1 and 3000 cm-1 and peaks at 2929.7 cm-1 and 1636.7 cm-1 were also observed.
.jpg)
Properties of Metronidazole tablets
The average weights of the tablets were between 490.60 and 494.90 mg (Table 3), which is within the official limit for tablets of 500 mg target weight.
.jpg)
Tablet hardness for all batches ranged between 38.44 and 73.26 N, although MTP prepared with Prosolv© was the hardest (73.26 N) of them. Increasing the amount of OKG in the co-processed excipients increased the hardness of the tablets (38.55<55.90=55.90 N) as observed for MT1, MT3, and MT5, respectively.
Tablet friability was between 0.82% and 1.54% for all the batches (Table 3). Those tablets prepared with the co-processed excipients (MT1, MT3, and MT5) were more friable (1.50%, 1.33%, and 1.41%, respectively) than MTC and MTP (0.82% and 0.94%, respectively). However, the Crushing Strength-Friability Ratio (CSFR) showed MT3 to be the strongest tablet with a value of 42.03, followed by 39.65 for MT5 and 25.63 for MT1. Values of MTC and MTP, on the other hand, had CSFR values of 31.20 and 77.93, respectively.
Tablet disintegration was between 33 and 111 s, with MT1, MT3, and MT5 disintegrating at 33.33, 33.83, and 63.87 s, respectively (Table 3). This condition portrays the ability of the co-processed excipients to elicit faster disintegration than Prosolv® (111.50 s). On the other hand, MT1 and MT3 had similar disintegration times (33.33 and 33.67 s, respectively) with MTC (33.67 s). Disintegration Efficiency Ratio (DER) values of MT1 and MT3 are 1.19 and 1.78, respectively, greater than the value 1 set as a lower limit for better disintegrating excipient. However, MT5 had a value of 0.89.
The fraction of drug released after 30 min, DE30 min, was lower in tablets containing the co-processed excipients than those containing the reference excipients (Table 3). However, MT1 had similar efficiency to MTC and MTP. At the end of 60 min dissolution, however, drug release from all the tablets was between 87.90% and 99.38% (Figure 2).
.jpg)
The rank order for drug release was MT1>MT5>MTP>MT3>MTC showing drug release from MT1 was higher than from similar tablet formulations (MT3 and MT5) or tablets prepared with reference excipients (MTC and MTP).
According to Table 4, f2 values are between 39.39% and 48.92% when the dissolution profile of tablets prepared with the co-processed (MT1, MT3, and MT5) was compared with those prepared with CombiLac© (MTC).
.jpg)
However, compared to tablets prepared with Prosolv® (MTP), the values fell between 47.22% and 65.67%.
Table 5 presents that the release kinetics for MT1, MT3, MT5, and MTP favors the Higuchi kinetics while that of MTC favors the first-order kinetics. However, the mechanism of drug release as determined by the Korsmeyer-Peppas model shows “n” values between 0.5836 and 0.6796, indicating that drug release was by Non-Fickian diffusion.
.jpg)
Discussion
The angle of repose reflects the ability of a powder to flow. It is measured by powder resistance to flow. Limits set for this measurement indicate that materials with angles ≤300 have excellent flow, those with values between 31º and 35o good flow, those between 36º and 40º fair flow, and those with values of ≥400 poor flow [26]. The presence of OKG in the co-processed excipients is observed to improve their flow.
Carr’s compressibility index indicates the propensity of powdered materials to decrease under pressure. The materials with values above 25% are cohesive and have poor flow. The results show that CYG excipients can be easily compressed under pressure with values between 11.66% and 19.58%. Similarly, the excipients were non-cohesive and possessed fair flow, as expressed by the Hausner ratio between 1.13 and 1.24 [27].
Okra gum (OKG) had higher hydration capacity and swelling ability attributable to the fact that gums like OKG have charged hydrogen atoms which attract water molecules resulting in marked swelling [28]. Increasing the concentration of OKG in the co-processed batches resulted in the increased swelling ability due to particle agglomeration and mechanical interlocking of the particles. This observation is in line with that of a recent study [29], where the addition of Okra gum to pregelatinized potato starch enhanced swelling of the co-processed product. Swelling is not only one mechanism of disintegration but is also a precursor to prolonged tablet disintegration due to the formation of a gelatinous layer around the tablets when it comes in contact with an aqueous medium [30].
FTIR spectra of metronidazole (Figure 1A) show characteristic peaks at 3205.5, indicating O-H stretching, at 3101.1 C=H stretch, while at 1535.7, the C=N vibration. The peaks representing 1472.3, 1423.3, and 1367.9 represent C=C aromatic ring vibrations, CH3 bending vibrations, and C-N stretch vibrations. In contrast, those around 1185.3, 1073.5, and 823.7 represent C-O stretch, C-O bond, and C-H aromatic vibrations which are consistent for metronidazole [31]. The spectra obtained for the co-processed excipient in Figure 1B are characteristic of the co-processed excipient and can identify the excipient. The spectra representing the combination of the co-processed excipient (Figure 1C) and metronidazole show some of the characteristic bonds of metronidazole. However, no new peaks were observed, indicating no interaction between the co-processed excipient and metronidazole.
The average weights of the tablets listed in Table 3 show none of the formulated tablets deviated from the average weight by 5% according to official specification [32]. Thus, the tablets have uniform weights. This finding shows that combining the co-processed excipients and metronidazole can be compressed under pressure without varying tablet weights.
Tablet hardness/crushing strength is a parameter that symbolizes the mechanical strength of the tablet. It reflects the property of a tablet to withstand transportation, storage, and handling [33]. An acceptable hardness for uncoated immediate release tablets is approximately 44.15 N [34]. However, this value could differ depending on the type of excipients used in the formulation. Hardness is associated with disintegration. If a tablet is too hard, it will not disintegrate or require a longer time to decompose. Soft tablets would disintegrate faster and would not withstand the stress of packaging or handling [35]. The results in Table 3 show that increasing the amount of OKG in CYG excipients increases the hardness of the tablets in the formulation in the order of MT1
Disintegration is the mechanical break-up of a tablet into particles upon contact with an aqueous medium. It is often associated with dissolution, where the drug is available for therapeutic action. Increasing the amount of OKG in the excipients prolongs the time taken for disintegration. The longer disintegration times observed with MT3 could be ascribed to the high mechanical strength of the batch because the stronger the tablets, the more difficult it becomes for the disintegration fluid to penetrate the tablets resulting in prolonged disintegration time.
Incorporation of the CYG excipients in the tablet formulations resulted in faster disintegration than those tablets prepared with a commercial superdisintegrant, Prosolv®. The case of the latter may be attributed to the low disintegration efficiency of microcrystalline cellulose and high tablet hardness, as observed in this study and also reported in other studies [40]. Disintegration times of MT1 and MT3 were similar to those of the co-processed multifunctional excipient CombiLac©. This result portrays the capability of the co-processed excipient (CYG) in effecting fast tablet disintegration, which is comparable with a previous study where prepared cross-linked starch disintegrates faster than Prosolv® [41] and that of Nnabuike and Oyeniyi [15] where Okra gum and cassava starch were co-processed. However, the finding was different from another study where similarly prepared co-processed excipients did not elicit tablet disintegration at the required official time [42].
Similar to crushing strength, the Disintegration Efficiency Ratio (DER) is an acceptable measure of the balance between the tablet’s strength and the disintegration property [43]. Values greater than 1 indicate that the investigated disintegrant has better property than the reference disintegrant. This effect shows that the co-processed excipients CYG1 and CYG3 have better disintegration properties than Prosolv®. In addition, the results show that CYG excipients can be used as a filler to bulk up tablet formulations; it possesses good binding ability by producing powerful tablets and fast tablet disintegrating ability.
Figure 2 shows the dissolution profile of the prepared metronidazole tablets. Dissolution efficiency at 30 min shows slower drug release from MT1, MT3, and MT5 than from MTC and MTP. However, at the end of the dissolution period, the rank order of drug release was MT1>MT5>MT3. Drug release from MT1 was higher than those of MT3 and MT5 and comparable to that of MTC but different from that of MTP.
Similarity fit factor (f2) is used to compare dissolution profiles of a test and reference formulation to indicate sameness or equivalence between both formulations. Values of f2 less than 50% suggest that the dissolution profiles of the compared formulations are different; values equal to 100 % depict identical profiles while those between 50% and 100% show similar profiles [25]. Table 4 presents all the values that were less than 50%, indicating differences in the dissolution profiles of the tablets prepared with the co-processed excipients (MT1, MT3, and MT5) and those prepared with CombiLac® (MTC). These differences could be attributed to the faster dissolution of MTC than the former formulations. On the other hand, only the profiles of MT3 and MT5 were similar to that of MTP prepared with Prosolv®; the dissolution profile of MT1 prepared with CYG1 varied from that of MTP as displayed in Figure 2.
The in vitro release data were fitted into the zero-order, first-order, Higuchi, and Korsmeyer-Peppas models. Then, the model with the highest coefficient was taken as the appropriate model to describe the possible mechanism of drug release. The results show Higuchi model had the highest co-efficient for MT1, MT3, MT5, and MTP. This finding implies a higher drug concentration in the tablet matrix than in the surrounding solution, even though drug diffusion is proposed to have occurred at a constant rate [44]. This result can explain why the drug release was slower at the initial stages of the dissolution studies for MT1, MT3, and MT5 than MTC. On the other hand, the highest co-efficient for MTC was the first order, where the rate of release was dependent on drug concentration within the tablet and could be the reason for the decline in drug release observed with MTC between 20 and 60 min. The release mechanism shows the exponent “n” values to be between 0.49 and 0.89, which indicates non-Fickian diffusion [41]. It suggests that drug release from the prepared tablets was controlled by swelling and diffusion mechanisms.
At the end of dissolution, CYG1 showed the fastest drug release, indicating a relationship between the disintegration of tablets containing CYG1 and dissolution. Quick disintegration increased the tablet dissolution rate by rapidly exposing the particles to the dissolution medium. In this study, drug release from all tablet formulations was within official specification [38] since the amount of metronidazole released was greater than 85%.
Conclusion
Co-processing Dioscorea rotundata starch and Abelmoschus esculentus fruit gum produced a multifunctional directly compressible excipient which could make robust tablets with superior disintegrant property to Prosolv®, a commercial superdisintegrant and comparable properties with CombiLac©, and a novel multifunctional excipient. This study shows the suitability of using cheap and readily available materials to produce a multifunctional excipient for direct compression of oral tablet formulations.
Ethical Considerations
Compliance with ethical guidelines
There were no ethical considerations to be considered in this research.
Funding
No external funding was received for this study. The authors provided the resources used for the study.
Authors' contributions
Conceptualization and study design: Olubunmi J. Olayemi; Investigation, data collection, and analysis: Judith E. John and Rashidat Abdullahi; Supervision and writing the original draft: Olubunmi J. Olayemi and Christianah Y. Isimi; Reviewing and final writing: Olubunmi J. Olayemi, Judith E. John, Rashidat Abdullahi, and Christianah Y. Isimi.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors thank the National Institute for Pharmaceutical Research and Development (NIPRD), Abuja, Nigeria, for providing the facilities to conduct this study.
References
- Apeji YE, Oyi AR, Isah AB, Allagh TS, Modi SR, Bansal AK. Development and optimization of a starch-based co-processed excipient for direct compression using mixture design. AAPS PharmSciTech. 2018; 19(2):866-80. [DOI:10.1208/s12249-017-0887-x] [PMID]
- Chougule AS, Dikpati A, Trimbake T. Formulation development techniques of co-processed excipients. J Adv Pharm Sci. 2012; 2(2):231-49. http://japsjournal.in/journals/2.Ajay_review_final_copy.pdf
- Mamatha B, Srilatha D, Sivanarayani CH, Desu PK, Venkateswara Rao P. Co-processed excipients: An overview. World J Pharm Res. 2017; 6(15):224-37. https://www.researchgate.net/publication/330193672
- Thulluru A, Madhavi C, Nandini K, Sirisha S, Spandana D. Co-processed excipients: New era in pharmaceuticals. Asian J Res Pharm Sci. 2019; 9(1):1-5. [DOI:10.5958/2231-5659.2019.00001.8]
- JRS Pharma. PROSOLV® SMCC: Silicified microcrystalline cellulose, PROSOLV® SMCC HD 90 [Internet]. 2021 [Updated 2021]. Available from: https://www.jrspharma.com/pharma_en/products-services/excipients/prosolv-smcc.php
- Saha S, Shahiwala AF. Multifunctional coprocessed excipients for improved tableting performance. Expert Opin Drug Deliv. 2009; 6(2):197-208. [DOI:10.1517/17425240802708978] [PMID]
- Colorcon. StarCap 1500® Fir Lit. Coloron. com/literature/marketing/StarCap.pdf. [Accessed: 7 July 2021]. https://www.colorcon.com/products-formulation/all-products/
- Ogunjimi AT, Alebiowu G. Flow and consolidation properties of neem gum coprocessed with two pharmaceutical excipients. Powder Technol. 2013; 246:187-92. [DOI:10.1016/j.powtec.2013.04.051]
- Pusapati RT, Kumar MK, Rapeti SS, Murthy T. Development of co-processed excipients in the design and evaluation of atorvastatin calcium tablets by direct compression method. Int J Pharm Investig. 2014; 4(2):102-6. [DOI:10.4103/2230-973X.133059] [PMID] [PMCID]
- Olowosulu AK, Oyi A, Isah AB, Ibrahim MA. Formulation and evaluation of novel coprocessed excipients of maize starch and acacia gum (StarAc) for direct compression tabletting. Int J Pharm Res Innov. 2011; 2:39-45. https://www.researchgate.net/publication/267368334
- Ayorinde JO, Odeniyi MA. Material and compression properties of native and co-processed breadfruit starches. Niger J Pharm Res. 2016; 12(1):21-9. https://www.ajol.info/index.php/njpr/article/view/163151
- Gandhi PP, Mundada AS. Formulation and evaluation of coprocessed excipient for mouth dissolving formulation. Int J Drug Dev Res. 2016; 8(1):30-5. https://www.ijddr.in/drug-development/formulation-and-evaluation-of-c=8924
- Bakre L, Osibajo D, Koiki G, Bamiro O. Material, compressional and tableting properties of ipomea batatas (sweet potato) starch co-processed with silicon dioxide. Acta Pharm Sci. 2019; 57(4):21-37. [DOI:10.23893/1307-2080.APS.05722]
- Okunlola A, Gbadamosi TA. Compaction and tableting behavior of a novel co-processed excipient in the formulation of metoprolol succinate tablets. Niger J Pharm Res. 2020; 16(2):127-42. [DOI:10.4314/njpr.v16i2.4]
- Nnabuike ND, James OY. Disintegration behavior of metronidazole tablets containing different ratios of co-processed Abelmoschus esculentus gum and Manihot esculenta starch. Int J Sci Res. 2019; 8(2):43-6. https://www.worldwidejournals.com/international-journal-of-scientific-research-=246
- Subhranshu P, Kumari CS, Sireesha G. Formulation and evaluation of metoprolol succinate orodispersible tablets using directly compressible coprocessed excipient of Moringa gum. Asian J Pham. 2020; 14(1):1-8. [DOI:10.22377/ajp.v14i1.3469]
- Adeoye O, Alebiowu G. Evaluation of coprocessed disintegrants produced from tapioca starch and mannitol in orally disintegrating paracetamol tablet. Acta Pol Pharm. 2014; 71(5):803-11. [PMID]
- Okoye EI, Onyekweli AO, Kunle OO. Okra gum- an economic choice for the amelioration of capping and lamination in tablets. Ann Biol Res. 2011; 2(2):30-42. https://www.researchgate.net/profile/Olobayo-Kunle/266867421
- Hussain A, Qureshi F, Abbas N, Arshad MS, Ali E. An evaluation of the binding strength of okra gum and the drug release characteristics of tablets prepared from it. Pharmaceutics. 2017; 9(2):20 [DOI:10.3390/pharmaceutics9020020] [PMID] [PMCID]
- Kemas UC, Nep EI, Agbowuro AA, Ochekpe NA. Effect of chemical modification on the proximate composition of M. esculentus starch and characterization using FTIR spectroscopy. World Pharm Res. 2012; 1(5):1234-49. https://www.researchgate.net/publication/283644993_EFFECT_OF_
- Somboonpanyakul P, Wang Q, Cui W, Barbut S, Jantawat P. Malva nut gum. (Part I): Extraction and physicochemical characterization. Carbohydr Polym. 2006; 64(2):247-53. [DOI:10.1016/j.carbpol.2005.11.034]
- Okunlola A. Flow, compaction and tabletting properties of co-processed excipients using pregelatinized Ofada rice starch and HPMC. J Excip Food Chem. 2018; 9(1):4-15. https://www.researchgate.net/publication/324795476
- Olayemi O, Oyi AR, Allagh TS. Comparative evaluation of maize, rice and wheat starch powders as pharmaceutical excipients. Nig J Pharm Sci. 2008; 7(1):131-8. https://www.researchgate.net/profile/Olubunmi-Olayemi
- Eraga SO, Ndukwe JO, Iwuagwu MA. An investigation of the direct compression properties of pre-gelatinized African bitter yam and cassava starches in acetylsalicylic acid tablet formulations. J Appl Sci Environ Manage. 2017; 21(5):855-62. [DOI:10.4314/jasem.v21i5.10]
- Anderson NH, Bauer M, Boussac N, Khan-Malek R, Munden P, Sardaro M. An evaluation of fit factors and dissolution efficiency for the comparison of in vitro dissolution profiles. J Pharm Biomed Anal. 1998; 17(4-5):811-22. [DOI:10.1016/S0731-7085(98)00011-9]
- Lumay G, Boschini F, Traina K, Bontempi S, Remy JC, Cloots R, et al. Measuring the flowing properties of powders and grains. Powder Technol. 2012; 224:19-27. [DOI:10.1016/j.powtec.2012.02.015]
- Reddy RS, Ramachandra CT, Hiregoudar Sh, Nidoni U, Ram J, Kammar M. Influence of processing conditions on functional and reconstitution properties of milk powder made from Osmanabadi goat milk by spray drying. Small Rumin Res. 2014; 119(1-3):130-7. [DOI:10.1016/j.smallrumres.2014.01.013]
- Omidian H, Park K. Hydrogels. In: Siepmann J, Siegel R, Rathbone M, editors. Fundamentals and Applications of Controlled Release Drug Delivery. Advances in Delivery Science and Technology. Boston, MA: Springer; 2012. pp. 75-105. [DOI:10.1007/978-1-4614-0881-9_4]
- Okunlola A. Preliminary characterization of co-processed excipients of okra (Abelmoschus esculentus) mucilage and pregelatinized potato starch. Niger J Pharm Res. 2021; 16(S):119-29. [DOI:10.4314/njpr.v16i2.14S]
- Siepmann J, Streubel A, Peppas NA. Understanding and predicting drug delivery from hydrophilic matrix tablets using the “sequential layer” model. Pharm Res. 2002; 19(3):306-14. [DOI:10.1023/A:1014447102710] [PMID]
- Patil PD, Desai SR, Disouza JI. Stability analysis and quantitative evaluation of metronidazole suspension. Indian J Pharm Biol Res. 2016; 4(3):5-14. https://www.proquest./openview/37e480c52652e64de083f3db693503b2/1.pdf
- British Pharmacopoeia Commission. British pharmacopoeia (BP) Vol. III. London: The Stationery Office Limited: London, UK. 2009. pp. 6578-85. https://www.google.com/books/edition/British_Pharmacopoeia/2GOYMwEACAAJ?hl=en
- Odeku OA, Itiola OA. Effects of interacting variables on the tensile strength and the release properties of paracetamol tablets. Trop J Pharm Res. 2003; 2(1):147-53. [DOI:10.4314/tjpr.v2i1.14579]
- Ministry of Health. Agência Nacional de Vigilância Sanitária (ANVISA). Brazilian Pharmacopeia, 5th Edition, Brasilia: Atheneu. 2010. https://www.gov.br/anvisa/pt-br/assuntos/farmacopeia/farmacopeia-brasileira/-file-1
- Markl D, Zeitler JA. A review of disintegration mechanisms and measurement techniques. Pharm Res. 2017; 34(5):890-917. [DOI:10.1007/s11095-017-2129-z] [PMID] [PMCID]
- Enauyatifard R, Azadbakht M, Fadakar Y. Assessment of Ferula gummosa gum as a binding agent in tablet formulations. Acta Pol Pharm. 2012; 69(2):291-8. [PMID]
- Gupta MM, Khoorban A, Ali A, Ramlogan O, Talukdar D. Comparative quality control study of different brands of diclofenac sodium tablet available in local and government pharmacies by in-vitro testing. Cureus. 2020; 12(11):e11348. [DOI:10.7759/cureus.11348]
- United States Pharmacopeia, 37th edition. https://www.usp.org/sites/default/files/usp/document/harmoniza.
- Njega EK, Maru SM, Tripo LJ. The binder effect of povidone on the mechanical properties of paracetamol containing tablets. East Cent Afr J Pharm Sci. 2018; 21(1-3):3-9. https://www.ajol.info/index.php/ecajps/article/view/186035
- Vodáčková P, Vraníková B, Svačinová P, Franc A, Elbl J, Muselik J, et al. Evaluation and comparison of three types of spray dried coprocessed excipient Avicel® for direct compression. Biomed Res Int. 2018; 2018:2739428. [DOI:10.1155/2018/2739428] [PMID] [PMCID]
- Gouda R, Baishya H, Qing Zh. Application of mathematical models in drug release kinetics of carbidopa and levodopa ER tablets. J Dev Drugs. 2017; 6(2):1000171. [DOI:10.4172/2329-6631.1000171]
- Ugoeze KC, Idris MEJ. Development of co-processed powders containing lactose, Mucuna flagellipes seed gum and Ipomoea batatas tuber starch. Int J Appl Biol Pharm Technol. 2020; 11(4):256-75. https://www.fortunejournals.com/articles/development-of-coprocessed-powders-c-.html
- Pachuau L, Dutta RS, Roy PK, Kalita P, Lalhlenmawia H. Physicochemical and disintegrant properties of glutinous rice starch of Mizoram, India. Int J Biol Macromol. 2017; 95:1298-304. [DOI:10.1016/j.ijbiomac.2016.11.029] [PMID]
- Dash S, Murthy PN, Nath L, Chowdhury P. Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm. 2010; 67(3):217-23. [PMID]
Type of Study: Original Research |
Subject:
Pharmaceutics
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |









