Volume 6, Issue 2 (2020)
Pharm Biomed Res 2020, 6(2): 133-142 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Adikwu E, Nnaedozie E. Protective Effect of Time-Modulated Cimetidine on Methotrexate-induced Liver Toxicity. Pharm Biomed Res 2020; 6 (2) :133-142
URL: http://pbr.mazums.ac.ir/article-1-267-en.html
URL: http://pbr.mazums.ac.ir/article-1-267-en.html
1- Department of Pharmacology and Toxicology, Faculty of Pharmacy, Niger Delta University, Bayelsa Stae, Nigeria.
2- Department of Pharmacology and Toxicology, Faculty of Pharmacy, Madonna University, Elele, Rivers State, Nigeria.
2- Department of Pharmacology and Toxicology, Faculty of Pharmacy, Madonna University, Elele, Rivers State, Nigeria.
Full-Text [PDF 1167 kb]
(1829 Downloads)
| Abstract (HTML) (5168 Views)
Effects on liver oxidative stress indices and histology
Liver SOD, CAT, GPX, GSH, and MDA levels were normal (P>0.05) in rats treated with CT. The liver levels of SOD, CAT, GPx, and GSH significantly (P<0.001) decreased whereas MDA significantly (P<0.001) increased in rats treated with MTX when compared to control (Table 3). In contrast, liver SOD, CAT, GPx, and GSH levels significantly increased whereas MDA levels significantly decreased in rats post-treated (P<0.05), co-treated (P<0.01), and pre-treated (P<0.001) with CT when compared with to MTX-treated rats (Table 3). Normal liver histology was observed in the control group (Figure 8A) whereas hepatocyte necrosis was observed in rats treated with MTX (Figure 8B). Also, the liver of rats post-treated with CT showed inflammatory cells (Figure 8C). The liver of rats pre-treated with CT showed ihepatocyte necrosis (Figure 8D) whereas the liver of rat co-treated with CT showed steatosis (Figure 8E).
Full-Text: (1710 Views)
Introduction
Methotrexate (MTX), an analog of folic acid, is one of the frequently used chemotherapeutic agents, especially in treating hematological malignancies and solid tumors. It is a well-established and effective treatment for different types of rheumatic diseases, severe psoriasis, and bowel diseases (like Crohn’s disease) [1]. Its anticancer activity involves the prevention of RNA and DNA syntheses through the inhibition of dihydrofolate reductase enzyme responsible for the conversion of folate to tetrahydrofolate [2]. Clinically, although the drug has some comparative advantages over some chemotherapeutic agents, its use is associated with hepatotoxicity [3]. Studies have reported a rate of 7% hepatotoxicity in cancer patients on MTX chemotherapy. Its hepatotoxic effect ranges from elevations in serum aminotransferases to changes in kidney histology [4]. Also, the use of high-dose MTX or prolonged use may result in hepatic fibrosis and cirrhosis [5]. The precise mechanism of MTX hepatotoxicity is unclear. However, it has been associated with increased activities of mediators of inflammation such as interleukin 1β, tumor necrosis factor α, and interleukin 6. Also, oxidative and nitrosative stress characterized by the production of free radicals and impaired redox balance have been reported. Furthermore, Lipid Peroxidation (LPO) which is a form of oxidative stress has been associated with MTX-induced hepatotoxicity [6].
Cimetidine (CT) is used for the management of patients with gastric ulcer and as prophylaxis for gastric aspiration syndrome. It is used during surgery to prevent stress-induced ulceration [7]. CT works by blocking H2 receptors on the stomach lining thereby preventing the interaction of histamine with H2 receptors, thus preventing acid secretion [8]. Besides its antihistamine effect, it has scavenging activities on monochloramine and hypochlorous acid, known as cytotoxic oxidants produced by inflammatory cells such as neutrophils. Also, CT can scavenge hydroxyl radicals, singlet oxygen, and peroxy radicals at a high constant rate [9]. This property can be used for the treatment of diseases characterized by free radical-mediated oxidative stress. Furthermore, studies suggest that it can be repurposed for the treatment of hepatotoxicity because it acts as an antidote for acetaminophen poisoning in rats [10]. Also, it inhibits hepatotoxicity caused by carbon tetrachloride (CCl4) in rats [11] and was effective in a rabbit model of rifampicin and isoniazid-induced hepatotoxicity [12]. Similarly, CT abrogates cocaine-induced hepatotoxicity in mice [13] and radiation-induced hepatotoxicity in rats [14].
The time of drug administration is crucial because it can affect therapeutic outcomes [15]. It has been reported that the trough to peak ratio of some drugs is directly related to the dosing time interval and individual response [16]. The time of medication administration is an important factor in the treatment or management of ailments and drug toxicities. It can affect the kinetics of a drug due to several factors, including circadian rhythms [17]. Timing in terms of post-treatment, pre-treatment, and co-treatment with xenobiotics have been used to abrogate or ameliorate toxicities caused by drugs and chemical agents [18, 19]. The best prospect in terms of therapeutic outcome considering the aforementioned treatments will probably depend on the kinetics of the drug and patient’s response. The current study assessed the protective effect of time-modulated CT on a rat model of MTX-induced hepatotoxicity.
Materials and Methods
Drugs
MTX was manufactured by Zuvius Lifesciences Ltd., (India), while CT was manufactured by Shandong Shenglu Pharmaceutical Company Ltd. (China). All other chemical substances used are of analytical grade. The aforementioned drugs were obtained from a pharmacy shop
Experimental animals
Adult male albino rats (200 g-250 g) were obtained from the experimental animal facility of the Department of Pharmacology and Toxicology, Faculty of Pharmacy, Madonna University, Elele, Rivers State, Nigeria. The rats were placed in cages (6 per cage) under natural light condition and temperature. The rats were fed with standard rodent chow and given water ad libitum and allowed for two weeks to acclimatize. This study was approved (Pharm/Res/No.33/ 2019) by the Research Ethics Committee of the Department of Pharmacology and Toxicology, Faculty of Pharmacy, Madonna University, Elele, Nigeria. The rats were handled based on the regulation of the European Parliament and Council on the Protection of Experimental Animals, 2010 [20].
Experimental protocol
Thirty-six adult albino rats were divided into 6 groups of 6 rats in each group.
The rats in group A (control) were injected intraperitoneally (IP) with one dose of 9% normal saline (0.2 mL) for 24 h. The rats in group B were treated IP with CT (20 mg/kg) [21] diluted with normal saline for 24 h. The rats in group C were treated IP with one dose of MTX (20 mg/kg) [22] dissolved in normal saline for 24 h. The rats in group D (pre-treatment group) were treated IP with CT (20 mg/kg) one hour before treatment with one dose of MTX (20 mg/kg) for 24 h. The rats in group E (co-administration group) were co-treated IP with CT (20 mg/kg) and MTX (20 mg/kg) for 24 h. The rats in group F (post-treatment group) were treated IP with one dose of MTX (20 mg/kg) one hour before treatment with one dose of CT (20 mg/kg) for 24 h.
Sacrifice of animals
Rats were sacrificed after drug treatment with diethyl ether, and blood samples were collected from the heart. The blood samples were centrifuged (1200g for 20 min) and serum samples were obtained for the evaluation of liver function indices. Liver samples were collected, weighed, and preserved for histological assessment. Also, liver samples were collected, washed in ice-cold 1.15% potassium chloride, and homogenized in 0.1 M Tris-HCl buffer, pH 7.4. The homogenates were centrifuged (1500g for 20 min) and the supernatants were extracted and evaluated for biochemical parameters.
Evaluation of biochemical parameters
Serum and liver gamma-glutamyl transferase (GGT), alkaline phosphatase (ALP), lactate dehydrogenase (LDH), alanine aminotransferase (ALT), conjugated bilirubin (CB), aspartate aminotransferase (AST), and total bilirubin (TB) levels were evaluated using commercial test kits (Randox Laboratories UK). Liver malondialdehyde (MDA) was assessed according to Buege and Aust study [23] and superoxide dismutase (SOD) as reported by Sun and Zigman [24]. Catalase (CAT) was evaluated according to Aebi [25] and glutathione peroxidase (GPx) according to Rotruck et al. [26]. Reduced glutathione (GSH) was evaluated as described by Sedlak and Lindsay [27] and total protein as described by Lowry et al. [28].
Histological examination of the liver
Liver samples were fixed in formal saline for 24 h, processed routinely, and embedded in paraffin blocks. Processed tissues were sectioned (3-5 μm thick), and slides were prepared and stained with hematoxylin and eosin. Stained sections were examined for histological changes under a light microscope.
Statistical analysis
Values are presented as Mean±SEM. Variables were statistically analyzed by one-way Analysis of Variance (ANOVA), followed by Tukey’s post hoc test. The significance level was set at P<0.05, P<0.01, and P<0.001 for different comparisons.
Results
Effects on the body, and liver weights and liver function parameters
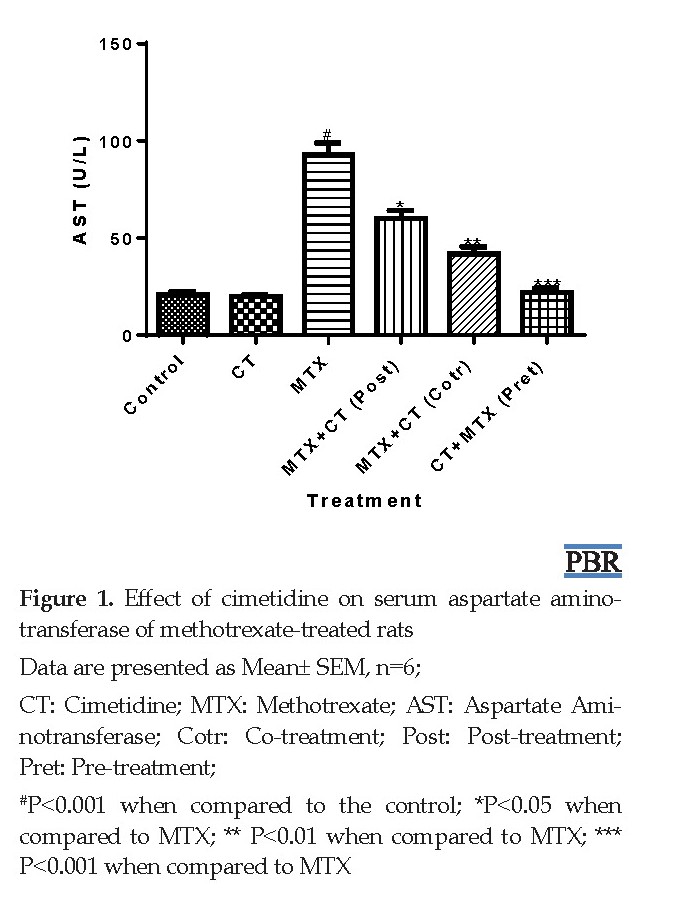
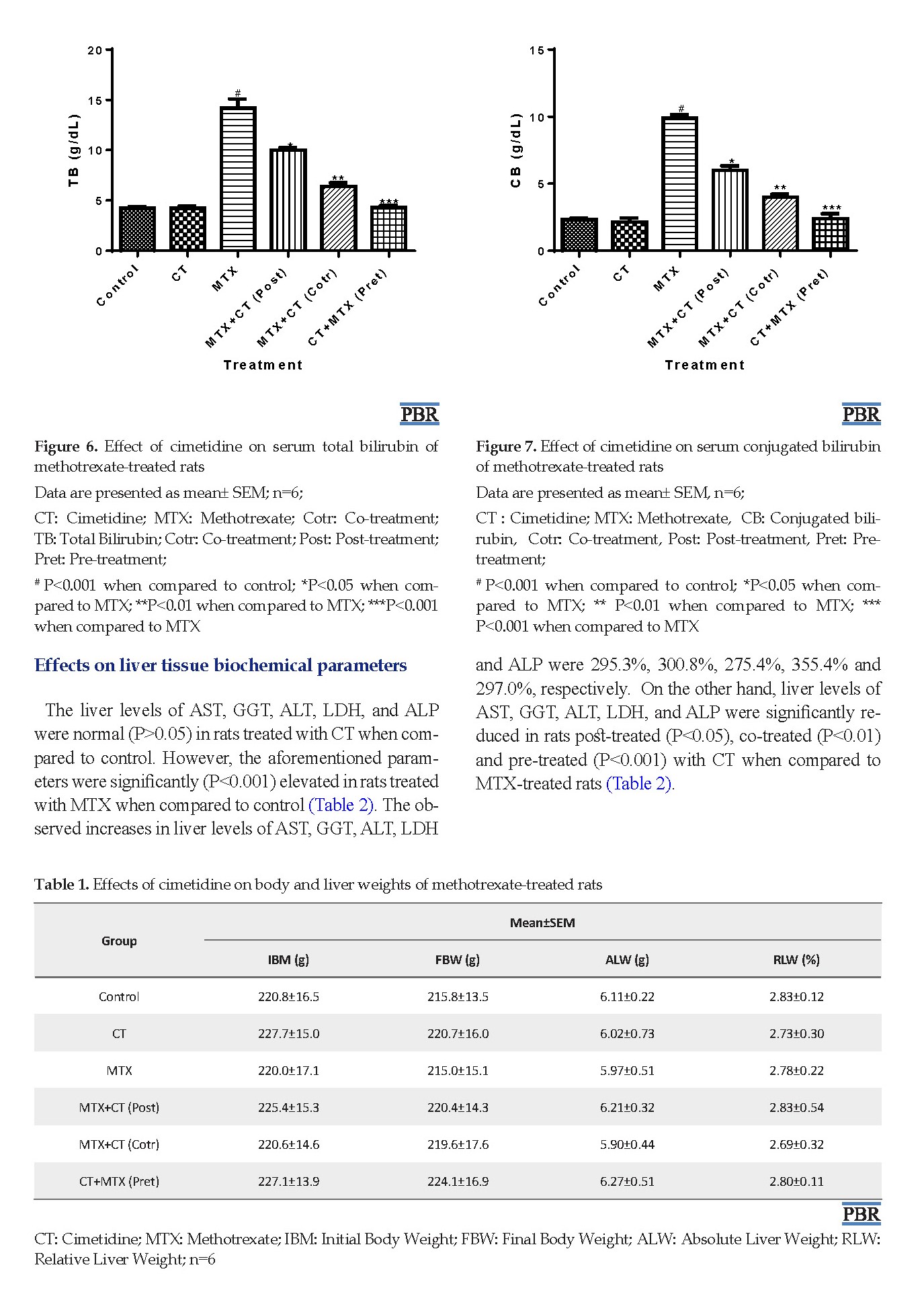
The body and liver weights, and serum TB, CB AST, GGT ALT, LDH, and ALP levels were normal (P>0.05) in CT-treated rats when compared to control. However, significant (P<0.001) elevations in serum TB, CB, AST, GGT, ALT, LDH, and ALP levels occurred in MTX-treated rats when compared to control (Table 1, Figures 1-8). The observed increases in the aforementioned parameters were 231.8%, 324.5%, 347.8%, 279.2%, 337.6%, 274.9%, and 315.1%, respectively. In contrast, serum TB, CB, AST, GGT, ALT, LDH, and ALP levels were significantly decreased rats in post-treated (P<0.05), co-treated (P<0.01), and pre-treated (P<0.001) with CT when compared to MTX-treated rats (Figures 1-8).
Methotrexate (MTX), an analog of folic acid, is one of the frequently used chemotherapeutic agents, especially in treating hematological malignancies and solid tumors. It is a well-established and effective treatment for different types of rheumatic diseases, severe psoriasis, and bowel diseases (like Crohn’s disease) [1]. Its anticancer activity involves the prevention of RNA and DNA syntheses through the inhibition of dihydrofolate reductase enzyme responsible for the conversion of folate to tetrahydrofolate [2]. Clinically, although the drug has some comparative advantages over some chemotherapeutic agents, its use is associated with hepatotoxicity [3]. Studies have reported a rate of 7% hepatotoxicity in cancer patients on MTX chemotherapy. Its hepatotoxic effect ranges from elevations in serum aminotransferases to changes in kidney histology [4]. Also, the use of high-dose MTX or prolonged use may result in hepatic fibrosis and cirrhosis [5]. The precise mechanism of MTX hepatotoxicity is unclear. However, it has been associated with increased activities of mediators of inflammation such as interleukin 1β, tumor necrosis factor α, and interleukin 6. Also, oxidative and nitrosative stress characterized by the production of free radicals and impaired redox balance have been reported. Furthermore, Lipid Peroxidation (LPO) which is a form of oxidative stress has been associated with MTX-induced hepatotoxicity [6].
Cimetidine (CT) is used for the management of patients with gastric ulcer and as prophylaxis for gastric aspiration syndrome. It is used during surgery to prevent stress-induced ulceration [7]. CT works by blocking H2 receptors on the stomach lining thereby preventing the interaction of histamine with H2 receptors, thus preventing acid secretion [8]. Besides its antihistamine effect, it has scavenging activities on monochloramine and hypochlorous acid, known as cytotoxic oxidants produced by inflammatory cells such as neutrophils. Also, CT can scavenge hydroxyl radicals, singlet oxygen, and peroxy radicals at a high constant rate [9]. This property can be used for the treatment of diseases characterized by free radical-mediated oxidative stress. Furthermore, studies suggest that it can be repurposed for the treatment of hepatotoxicity because it acts as an antidote for acetaminophen poisoning in rats [10]. Also, it inhibits hepatotoxicity caused by carbon tetrachloride (CCl4) in rats [11] and was effective in a rabbit model of rifampicin and isoniazid-induced hepatotoxicity [12]. Similarly, CT abrogates cocaine-induced hepatotoxicity in mice [13] and radiation-induced hepatotoxicity in rats [14].
The time of drug administration is crucial because it can affect therapeutic outcomes [15]. It has been reported that the trough to peak ratio of some drugs is directly related to the dosing time interval and individual response [16]. The time of medication administration is an important factor in the treatment or management of ailments and drug toxicities. It can affect the kinetics of a drug due to several factors, including circadian rhythms [17]. Timing in terms of post-treatment, pre-treatment, and co-treatment with xenobiotics have been used to abrogate or ameliorate toxicities caused by drugs and chemical agents [18, 19]. The best prospect in terms of therapeutic outcome considering the aforementioned treatments will probably depend on the kinetics of the drug and patient’s response. The current study assessed the protective effect of time-modulated CT on a rat model of MTX-induced hepatotoxicity.
Materials and Methods
Drugs
MTX was manufactured by Zuvius Lifesciences Ltd., (India), while CT was manufactured by Shandong Shenglu Pharmaceutical Company Ltd. (China). All other chemical substances used are of analytical grade. The aforementioned drugs were obtained from a pharmacy shop
Experimental animals
Adult male albino rats (200 g-250 g) were obtained from the experimental animal facility of the Department of Pharmacology and Toxicology, Faculty of Pharmacy, Madonna University, Elele, Rivers State, Nigeria. The rats were placed in cages (6 per cage) under natural light condition and temperature. The rats were fed with standard rodent chow and given water ad libitum and allowed for two weeks to acclimatize. This study was approved (Pharm/Res/No.33/ 2019) by the Research Ethics Committee of the Department of Pharmacology and Toxicology, Faculty of Pharmacy, Madonna University, Elele, Nigeria. The rats were handled based on the regulation of the European Parliament and Council on the Protection of Experimental Animals, 2010 [20].
Experimental protocol
Thirty-six adult albino rats were divided into 6 groups of 6 rats in each group.
The rats in group A (control) were injected intraperitoneally (IP) with one dose of 9% normal saline (0.2 mL) for 24 h. The rats in group B were treated IP with CT (20 mg/kg) [21] diluted with normal saline for 24 h. The rats in group C were treated IP with one dose of MTX (20 mg/kg) [22] dissolved in normal saline for 24 h. The rats in group D (pre-treatment group) were treated IP with CT (20 mg/kg) one hour before treatment with one dose of MTX (20 mg/kg) for 24 h. The rats in group E (co-administration group) were co-treated IP with CT (20 mg/kg) and MTX (20 mg/kg) for 24 h. The rats in group F (post-treatment group) were treated IP with one dose of MTX (20 mg/kg) one hour before treatment with one dose of CT (20 mg/kg) for 24 h.
Sacrifice of animals
Rats were sacrificed after drug treatment with diethyl ether, and blood samples were collected from the heart. The blood samples were centrifuged (1200g for 20 min) and serum samples were obtained for the evaluation of liver function indices. Liver samples were collected, weighed, and preserved for histological assessment. Also, liver samples were collected, washed in ice-cold 1.15% potassium chloride, and homogenized in 0.1 M Tris-HCl buffer, pH 7.4. The homogenates were centrifuged (1500g for 20 min) and the supernatants were extracted and evaluated for biochemical parameters.
Evaluation of biochemical parameters
Serum and liver gamma-glutamyl transferase (GGT), alkaline phosphatase (ALP), lactate dehydrogenase (LDH), alanine aminotransferase (ALT), conjugated bilirubin (CB), aspartate aminotransferase (AST), and total bilirubin (TB) levels were evaluated using commercial test kits (Randox Laboratories UK). Liver malondialdehyde (MDA) was assessed according to Buege and Aust study [23] and superoxide dismutase (SOD) as reported by Sun and Zigman [24]. Catalase (CAT) was evaluated according to Aebi [25] and glutathione peroxidase (GPx) according to Rotruck et al. [26]. Reduced glutathione (GSH) was evaluated as described by Sedlak and Lindsay [27] and total protein as described by Lowry et al. [28].
Histological examination of the liver
Liver samples were fixed in formal saline for 24 h, processed routinely, and embedded in paraffin blocks. Processed tissues were sectioned (3-5 μm thick), and slides were prepared and stained with hematoxylin and eosin. Stained sections were examined for histological changes under a light microscope.
Statistical analysis
Values are presented as Mean±SEM. Variables were statistically analyzed by one-way Analysis of Variance (ANOVA), followed by Tukey’s post hoc test. The significance level was set at P<0.05, P<0.01, and P<0.001 for different comparisons.
Results
Effects on the body, and liver weights and liver function parameters
The body and liver weights, and serum TB, CB AST, GGT ALT, LDH, and ALP levels were normal (P>0.05) in CT-treated rats when compared to control. However, significant (P<0.001) elevations in serum TB, CB, AST, GGT, ALT, LDH, and ALP levels occurred in MTX-treated rats when compared to control (Table 1, Figures 1-8). The observed increases in the aforementioned parameters were 231.8%, 324.5%, 347.8%, 279.2%, 337.6%, 274.9%, and 315.1%, respectively. In contrast, serum TB, CB, AST, GGT, ALT, LDH, and ALP levels were significantly decreased rats in post-treated (P<0.05), co-treated (P<0.01), and pre-treated (P<0.001) with CT when compared to MTX-treated rats (Figures 1-8).
Effects on liver tissue biochemical parameters
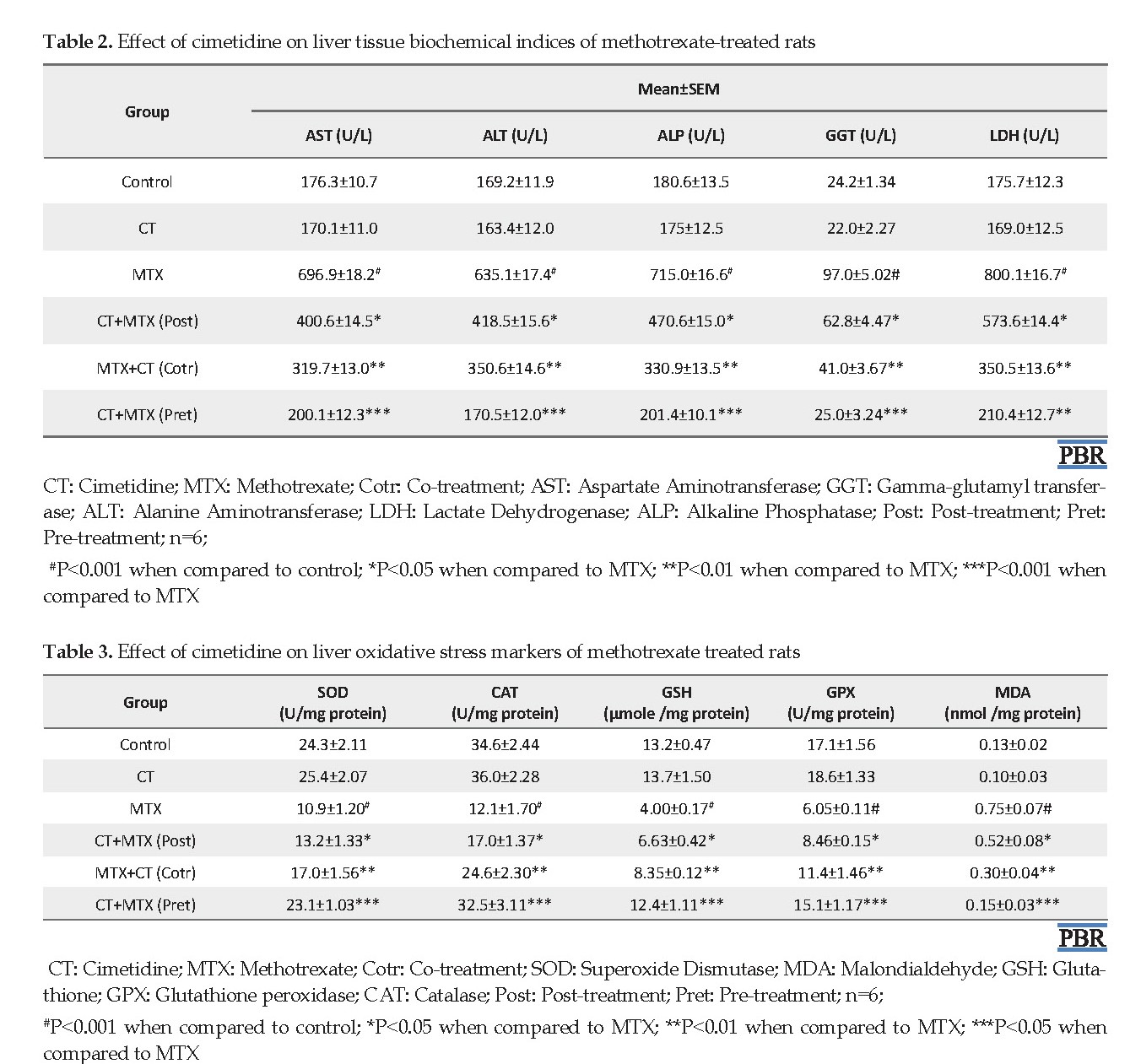
The liver levels of AST, GGT, ALT, LDH, and ALP were normal (P>0.05) in rats treated with CT when compared to control. However, the aforementioned parameters were significantly (P<0.001) elevated in rats treated with MTX when compared to control (Table 2). The observed increases in liver levels of AST, GGT, ALT, LDH and ALP were 295.3%, 300.8%, 275.4%, 355.4% and 297.0%, respectively. On the other hand, liver levels of AST, GGT, ALT, LDH, and ALP were significantly reduced in rats post-treated (P<0.05), co-treated (P<0.01) and pre-treated (P<0.001) with CT when compared to MTX-treated rats (Table 2).
The liver levels of AST, GGT, ALT, LDH, and ALP were normal (P>0.05) in rats treated with CT when compared to control. However, the aforementioned parameters were significantly (P<0.001) elevated in rats treated with MTX when compared to control (Table 2). The observed increases in liver levels of AST, GGT, ALT, LDH and ALP were 295.3%, 300.8%, 275.4%, 355.4% and 297.0%, respectively. On the other hand, liver levels of AST, GGT, ALT, LDH, and ALP were significantly reduced in rats post-treated (P<0.05), co-treated (P<0.01) and pre-treated (P<0.001) with CT when compared to MTX-treated rats (Table 2).
Effects on liver oxidative stress indices and histology
Liver SOD, CAT, GPX, GSH, and MDA levels were normal (P>0.05) in rats treated with CT. The liver levels of SOD, CAT, GPx, and GSH significantly (P<0.001) decreased whereas MDA significantly (P<0.001) increased in rats treated with MTX when compared to control (Table 3). In contrast, liver SOD, CAT, GPx, and GSH levels significantly increased whereas MDA levels significantly decreased in rats post-treated (P<0.05), co-treated (P<0.01), and pre-treated (P<0.001) with CT when compared with to MTX-treated rats (Table 3). Normal liver histology was observed in the control group (Figure 8A) whereas hepatocyte necrosis was observed in rats treated with MTX (Figure 8B). Also, the liver of rats post-treated with CT showed inflammatory cells (Figure 8C). The liver of rats pre-treated with CT showed ihepatocyte necrosis (Figure 8D) whereas the liver of rat co-treated with CT showed steatosis (Figure 8E).
Discussion
This study assessed the protective effect of time-modulated cimetidine against MTX-induced hepatotoxicity in a rat model. An important requirement in toxicological experiments is the assessments of the effects of xenobiotics on organ and body weights [29]. In this study, the induction of hepatotoxicity with MTX did not alter body and liver weights. Serum aminotransferases (AST and ALT) are used for differential diagnosis of liver disease and higher levels show liver dysfunction [30]. Also, serum activities of CT, TB, LDH, and GGT correlate with liver function and elevated activities are observed in hepatic disorders [31]. The serum and liver activities of CT, CB, AST, GGT, ALT, LDH, and ALP were high in rats treated with MTX. This finding agrees with previous studies [32]. The elevations in the activities of the aforementioned indexes are clear signs of hepatic injury [33]. However, the activities of CB, TB, AST, GGT, ALT, LDH, and ALP decreased in rats’ pre-treated, co-treated, and post-treated with CT. The decreases in the activities of the aforementioned parameters were most seen in rats pre-treated with CT.
This study assessed the protective effect of time-modulated cimetidine against MTX-induced hepatotoxicity in a rat model. An important requirement in toxicological experiments is the assessments of the effects of xenobiotics on organ and body weights [29]. In this study, the induction of hepatotoxicity with MTX did not alter body and liver weights. Serum aminotransferases (AST and ALT) are used for differential diagnosis of liver disease and higher levels show liver dysfunction [30]. Also, serum activities of CT, TB, LDH, and GGT correlate with liver function and elevated activities are observed in hepatic disorders [31]. The serum and liver activities of CT, CB, AST, GGT, ALT, LDH, and ALP were high in rats treated with MTX. This finding agrees with previous studies [32]. The elevations in the activities of the aforementioned indexes are clear signs of hepatic injury [33]. However, the activities of CB, TB, AST, GGT, ALT, LDH, and ALP decreased in rats’ pre-treated, co-treated, and post-treated with CT. The decreases in the activities of the aforementioned parameters were most seen in rats pre-treated with CT.
Oxidative Stress (OS) is a condition that favors oxidant generation and depletion of antioxidants leading to tissue injury [34]. In the current study, we observed decreases in liver antioxidant (CAT, GPX SOD, and GSH) activities in MTX-treated rats. This observation is a sign of OS which is consistent with earlier studies [35]. However, marked up-regulation in the activities of liver antioxidants were noted in rats’ pre-treated, co-treated, and post-treated with CT. MDA is a metabolite of free radical-induced LPO cascade. It is an essential indicator of OS and the breakdown of lipid layers [36]. The current study observed elevated MDA level in MTX-treated rats which is an indication of LPO that is consistent with earlier reports [37]. However, decreased LPO characterized by low levels of MDA were noted in rats’ pre-treated, co-treated, and post-treated with CT. The lowest level of MDA was observed in rats pre-treated with CT. The current study showed hepatocyte necrosis in MTX-treated rats which is consistent with previous findings [38].
Pre-treatment, offered, but co-treatment, and post-treatment with CT did not offer significant protection against MTX-induced hepatocyte necrosis. The exact mechanism of MTX-induced hepatotoxicity is not clear, but some mechanisms have been proposed. These mechanisms include direct toxic effect on the liver [39], inhibition of DNA synthesis, mitochondria damage, OS, and decreased hepatic folate levels by MTX polyglutamates [40]. The prevention of MTX-induced hepatotoxicity by CT might be due to its inhibitory effect on the above-mentioned mechanisms. CT has oxygen-radical scavenging activity. It scavenges hydroxyl radicals [41] and superoxide anions thereby preventing OS and LPO [42, 43]. Also, it can inhibit cytochrome P-450 monooxygenation reactions, which are associated with the production of toxic oxygen species [44]. Furthermore, the kinetics of CT in humans is similar to that of rats. It reaches peak plasma concentration in 45-75 minutes after administration resulting in blood levels averaging 2.8 μmol/L (0.7 mg/L). Its maximum activity is achieved when blood levels exceeded 0.5 mg/L [45]. Therefore, pre-treatment with CT produces the best hepatoprotective activity probably because its peak plasma concentration was attained before treatment with MTX. CT might have inhibited the activity of MTX before reaching its hepatic target. Furthermore, MTX undergoes hepatic and intracellular metabolism to polyglutamated forms, which are essential for its pharmacological and hepatotoxic activity [45]. Studies have shown that CT can inhibit the hepatic microsomal oxidation system [46]. Therefore, at peak plasma concentration, pre-treatment with CT might have inhibited the hepatic biotransformation of MTX by hepatic microsomal oxidation system leading to reduced production of its polyglutamated forms.
Pre-treatment, co-treatment, and post-treatment with CT attenuate MTX-induced hepatotoxicity, but the maximum effect was seen in rats pre-treated with CT.
Ethical Considerations
Compliance with ethical guidelines
The directive (2010/63/EU) of the European :union: Parliament on the handling of laboratory animals for scientific purposes was used for this study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
Conceptualization, original draft preparation, data curation, manuscript review and editing, and data analysis: Elias Adikwu; Original draft preparation, data curation, manuscript review and editing, data analysis, animal handling: Emmanuel Nnaedozie.
Conflict of interest
The authors declare no conflict of interest.
References
Babiak RM, Campello AP, Carnieri EG, Oliveira MB. Methotrexate: Pentose cycle and oxidative stress. Cell Biochem Funct. 1998; 16(4):283-93. [DOI:10.1002/(SICI)1099-0844(1998120)16:4<283::AID-CBF801>3.0.CO;2-E] [PMID]
Rajagopalan PTR, Zhiquan Z, McCourt L, Dwyer M, J Benkovic ST, Hammes GG. Interaction of dihydrofolate reductase with methotrexate: Ensemble and single-molecule kinetics. Proc Natl Acad Sci U S A. 2002; 99(21):13481-6. [DOI:10.1073/pnas.172501499] [PMID] [PMCID]
Tunali-Akbay T, Sehirli O, Ercan F, Sener G. Resveratrol protects against methotrexate-induced hepatic injury in rats. J Pharm Pharm Sci. 2010; 13(2):303-10. [DOI:10.18433/J30K5Q] [PMID]
Hytiroglou P, Tobias H, Saxena R, Abramidou M, Papadimitriou CS, Theise ND. The canals of hearing might represent a target of methotrexate hepatic toxicity. Am J Clin Pathol. 2014; 121(3):324-9. [DOI:10.1309/5HR90TNC4Q4JRXWX] [PMID]
Patel NN, Ghodasara DJ, Pandey S, Ghodasara PD, Khorajiya JH, Joshi BP, et al. Subacute toxicopathological studies of methotrexate in Wistar rats. Veter World. 2014; 7(7):489-95. [DOI:10.14202/vetworld.2014.489-495]
Samdanci ET, Huz M, Ozhan O, Tanbek K, Pamukcu E, Akatli AN, Parlakpinar H. Cytoprotective effects of molsidomine against methotrexate-induced hepatotoxicity: an experimental rat study. Drug Des Devel Ther. 2019; 13:13-21.
Strain JD, Moore EE, Markovchick VJ, Duzer-Moore SV. Cimetidine for the prophylaxis of potential gastric acid aspiration pneumonitis in trauma patients. J Trau. 1981; 21(1):49-51. [DOI:10.1097/00005373-198101000-00010] [PMID]
Singh H, Pahwa S, Dhamija K, Arora V. Cimetidine: A review. Int J Chem Tech Res. 2018; 11(2):115-23. http://www.sphinxsai.com/2018/ch_vol11_no2/abstracts/A(115-123)V11N02CT.pdf
Lapenna D, De Gioia S, Mezzetti A, Grossi L, Festi D, Marzio L, et al. H2-receptor antagonists are scavengers of oxygen radicals. Eur J Clin Invest. 1994; 24(7):476-81. [DOI:10.1111/j.1365-2362.1994.tb02378.x] [PMID]
Rolband GC, Marcuard SP. Cimetidine in the treatment of acetaminophen overdose. J Clin Gastroenterol. 1991; 13(1):79-82. [DOI:10.1097/00004836-199102000-00017] [PMID]
Nedelcu L, Șandor V, Dumitrașcu DL. Protective effect of cimetidine in carbon tetrachloride-treated rats. Biomed Res. 2019; 30(2):303-7 [DOI:10.35841/biomedicalresearch.30-19-063]
Kalra BS, Aggarwal S, Khurana N, Gupta U. Effect of cimetidine on hepatotoxicity induced by isoniazid-rifampicin combination in rabbits Bhupinder. Indian J Gastroenterol. 2007; 26(1):18-21. [PMID]
Peterson FJ, Knodell RG, Lindemann NJ, Steele NM. Prevention of acetaminophen and cocaine hepatotoxicity in mice by cimetidine treatment. Gastroenterology. 1983; 85(1):122-9. [DOI:10.1016/S0016-5085(83)80238-8] [PMID]
Jiang DW, Wang QR, Shen XR, He Y, Qian TT, Liu Q, et al. Radioprotective effects of cimetidine on rats irradiated by long-term, low-dose-rate neutrons and 60 Co γ-rays. Mil Med Res. 2017; 4:7. [DOI:10.1186/s40779-017-0116-7] [PMID] [PMCID]
Kaur G, Gan YL, Phillips CL, Wong K, Saini B. Chronotherapy in practice: The perspective of the community pharmacist. Int J Clin Pharm. 2016; 38(1):171-82. [DOI:10.1007/s11096-015-0228-7] [PMID]
Elliot HL, Meredith PA. Calculation of trough to peak ratio in a research unit setting advantages and disadvantages. Am J Hypertens. 1996; 9(10):71S-75S. [DOI:10.1016/S0895-7061(96)00266-X]
Ballesta A, Innominato PF, Dallmann R, Rand DA, Lévi FA. Systems chronotherapeutics. Pharmacol Rev. 2017; 69(2):161-99. [DOI:10.1124/pr.116.013441] [PMID] [PMCID]
Adikwu E, Braimbaifa N, Obianime AW. Melatonin and alpha lipoic acid: Possible mitigants for lopinavir/ritonavir- induced renal toxicity in male albino rats. Physiol Pharmacol. 2015; 19(4):232-40.
Nathiya S, Rajaram S, Abraham P. Hesperidin alleviates antitubercular drug induced oxidative stress, inflammation and apoptosis in rat liver. Int J Biomed Res. 2016; 7(7):439-46. [DOI:10.7439/ijbr.v7i7.3414]
The European Parliament And The Council Of The European :union:. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. Official Journal of the European :union:. 2010. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32010L0063
Al-Jawad FH, Abdulsahib WK, Al-Attar Z, Al-Hussaini JA. Protective effect of cimetidine, isosorbide dinitrate & vitamin c in experimental model of acute liver injury. Sch Acad J Pharm. 2018; 7(7):308-12. http://saspublisher.com/wp-content/uploads/2018/07/SAJP-77-308-312-c.pdf
Dalaklioglu S, Genc GE, Aksoy NH, Akcit F, Gumuslu S. Resveratrol ameliorates methotrexate-induced hepatotoxicity in rats via inhibition of lipid peroxidation. Hum Exp Toxicol. 2013; 32(6):662-71. [DOI:10.1177/0960327112468178] [PMID]
Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol. 1978; 52:302-10. [DOI:10.1016/S0076-6879(78)52032-6] [PMID]
Sun M, Zigma S. An improved spectrophotometer assay of superoxide dismutase based on epinephrine antioxidation. Anal Biochem. 1978; 90(1):81-9. [DOI:10.1016/0003-2697(78)90010-6] [PMID]
Aebi H. Catalase in vitro. Meth in Enzymol. 1984; 105:121- 6. [DOI:10.1016/S0076-6879(84)05016-3]
Rotruck JT, Rope AL, Ganther HF, Swason AB, Hafeman DG, Hoekstra WG. Selenium: Biochemical role as a component of glutathione peroxidase. Science. 1973; 179(4073):588-90. [DOI:10.1126/science.179.4073.588] [PMID]
Sedlak J, Lindsay RH. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem. 1968; 25:192-205. [DOI:10.1016/0003-2697(68)90092-4] [PMID]
Gornall AG, Bardawill CJ, David MM. Determination of serum proteins by means of the biureto reaction. J Biol Chem. 1949; 177:751-66. https://www.jbc.org/content/177/2/751.short
Bailey SA, Zidell RH, Perry RW. Relationships between organ weight and body/brainweight in the rat: What is the best analytical endpoint? Toxicol Pathol. 2004; 32(4):448-66.[DOI:10.1080/01926230490465874] [PMID]
Vroon DH, Israili Z. Aminotransferases. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The history, physical, and laboratory examinations. Boston: Butterworths; 1990. [PMID]
Elias SES, Eltom A, Osman AL, Babker AMA. Gamma glutamyl transferase and lactate dehydrogenase as biochemical markers of severity of preeclampsia among Sudanese pregnant women. Int J Reprod Contracept Obstet Gynecol. 2018; 7(8):3020-3. [DOI:10.18203/2320-1770.ijrcog20183294]
Hadi NR, Al-Amran FG, Swadi A. Metformin ameliorates methotrexate-induced hepatotoxicity. J Pharmacol Pharmacother. 2012; 3(3):248-53. [DOI:10.4103/0976-500X.99426] [PMID] [PMCID]
Tag HM. Hepatoprotective effect of mulberry (Morus nigra) leaves extract against methotrexate induced hepatotoxicity in male albino rat. BMC Comp and Altern Med. 2015; 15:252. [DOI:10.1186/s12906-015-0744-y] [PMID] [PMCID]
Halliwell B. Free radicals, antioxidants and human disease: Curiosity, cause or consequence? Lancet. 1994; 344(8924):721-4. [DOI:10.1016/S0140-6736(94)92211-X] [PMID]
Jahovic N, Cevik H, Sehirli OA, Yeğen BC, Sener G. Melatonin prevents methotrexate - induced hepatorenal oxidative injury in rats. J Pineal Res. 2003; 34(4):282-7. [DOI:10.1034/j.1600-079X.2003.00043.x] [PMID]
Gasparovic AC, Jaganjac M, Mihaljevic B, Sunjic SB, Zarkovic N. Assays for the measurement of lipid peroxidation.Methods Mol Biol. 2013; 965:283-96. [DOI:10.1007/978-1-62703-239-1_19] [PMID]
Kose E, Sapmaz HI, Sarihan E, Vardi N, Turkoz Y, Ekinci N. Beneficial effects of montelukast against methotrexate-induced liver toxicity: A biochemical and histological study. ScientificWorldJournal. 2012; 2012:987508. [DOI:10.1100/2012/987508] [PMID] [PMCID]
Soliman ME. Evaluation of the possible protective role of folic acid on the liver toxicity induced experimentally by methotrexate in adult male albino rats Egypt. J Histol. 2009; 1(1):118-28. https://www.sid.ir/en/journal/ViewPaper.aspx?ID=430087
Ros S, Juanola X, Condom E, Canas C, Riera J, Guardiola J, et al. Light and electron microscopic analysis of liver biopsy samples from rheumatoid arthritis patients receiving long-term methotrexate therapy. Scand J Rheumatol. 2002; 3(6):330-6. [DOI:10.1080/030097402320817040] [PMID]
Saka S, Aouacheri O. The investigation of the oxidative stressrelated parameters in high doses methotrexate-induced albino wistar rats. J Bioequiv Availab. 2017; 9(2):372-6. https://bit.ly/3dr7TYZ
Uchida K, Kawakishi S. Cimetidine anti-ulcer drug as a powerful hydroxyl radical scavenger. Agric and Biol Chem. 1990; 54(9):2485-7. [DOI:10.1271/bbb1961.54.2485]
Číž M, Lojek A. Modulation of neutrophil oxidative burst via histamine receptors. Br J Pharmacol. 2013; 170(1):17-22. [DOI:10.1111/bph.12107] [PMID] [PMCID]
Lambat Z, Limson JL, Daya S. Cimetidine: Antioxidant and metal‐binding properties. J Pharm Pharmacol. 2002; 54(12):1681-6. [DOI:10.1111/j.2042-7158.2002.tb02404.x] [PMID]
Shaik IH, Mehvar R. Effects of cytochrome p450 inhibition by cimetidine on the warm hepatic ischemia-reperfusion injury in rats. J Surg Res. 2010; 159(2):680-8. [DOI:10.1016/j.jss.2008.09.016] [PMID]
Furst DE, Kremer JM. Methotrexate in rheumatoid arthritis. Arthritis Rheum. 1988; 31(3):305-14. [DOI:10.1002/art.1780310301] [PMID]
Singh H, Pahwa S., Dhamija K, Arora V Cimetidine: A review. Int J Chem Tech Res. 2018; 11(02):115-23. [DOI:10.20902/IJCTR.2018.110944]
Pre-treatment, co-treatment, and post-treatment with CT attenuate MTX-induced hepatotoxicity, but the maximum effect was seen in rats pre-treated with CT.
Ethical Considerations
Compliance with ethical guidelines
The directive (2010/63/EU) of the European :union: Parliament on the handling of laboratory animals for scientific purposes was used for this study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
Conceptualization, original draft preparation, data curation, manuscript review and editing, and data analysis: Elias Adikwu; Original draft preparation, data curation, manuscript review and editing, data analysis, animal handling: Emmanuel Nnaedozie.
Conflict of interest
The authors declare no conflict of interest.
References
Babiak RM, Campello AP, Carnieri EG, Oliveira MB. Methotrexate: Pentose cycle and oxidative stress. Cell Biochem Funct. 1998; 16(4):283-93. [DOI:10.1002/(SICI)1099-0844(1998120)16:4<283::AID-CBF801>3.0.CO;2-E] [PMID]
Rajagopalan PTR, Zhiquan Z, McCourt L, Dwyer M, J Benkovic ST, Hammes GG. Interaction of dihydrofolate reductase with methotrexate: Ensemble and single-molecule kinetics. Proc Natl Acad Sci U S A. 2002; 99(21):13481-6. [DOI:10.1073/pnas.172501499] [PMID] [PMCID]
Tunali-Akbay T, Sehirli O, Ercan F, Sener G. Resveratrol protects against methotrexate-induced hepatic injury in rats. J Pharm Pharm Sci. 2010; 13(2):303-10. [DOI:10.18433/J30K5Q] [PMID]
Hytiroglou P, Tobias H, Saxena R, Abramidou M, Papadimitriou CS, Theise ND. The canals of hearing might represent a target of methotrexate hepatic toxicity. Am J Clin Pathol. 2014; 121(3):324-9. [DOI:10.1309/5HR90TNC4Q4JRXWX] [PMID]
Patel NN, Ghodasara DJ, Pandey S, Ghodasara PD, Khorajiya JH, Joshi BP, et al. Subacute toxicopathological studies of methotrexate in Wistar rats. Veter World. 2014; 7(7):489-95. [DOI:10.14202/vetworld.2014.489-495]
Samdanci ET, Huz M, Ozhan O, Tanbek K, Pamukcu E, Akatli AN, Parlakpinar H. Cytoprotective effects of molsidomine against methotrexate-induced hepatotoxicity: an experimental rat study. Drug Des Devel Ther. 2019; 13:13-21.
Strain JD, Moore EE, Markovchick VJ, Duzer-Moore SV. Cimetidine for the prophylaxis of potential gastric acid aspiration pneumonitis in trauma patients. J Trau. 1981; 21(1):49-51. [DOI:10.1097/00005373-198101000-00010] [PMID]
Singh H, Pahwa S, Dhamija K, Arora V. Cimetidine: A review. Int J Chem Tech Res. 2018; 11(2):115-23. http://www.sphinxsai.com/2018/ch_vol11_no2/abstracts/A(115-123)V11N02CT.pdf
Lapenna D, De Gioia S, Mezzetti A, Grossi L, Festi D, Marzio L, et al. H2-receptor antagonists are scavengers of oxygen radicals. Eur J Clin Invest. 1994; 24(7):476-81. [DOI:10.1111/j.1365-2362.1994.tb02378.x] [PMID]
Rolband GC, Marcuard SP. Cimetidine in the treatment of acetaminophen overdose. J Clin Gastroenterol. 1991; 13(1):79-82. [DOI:10.1097/00004836-199102000-00017] [PMID]
Nedelcu L, Șandor V, Dumitrașcu DL. Protective effect of cimetidine in carbon tetrachloride-treated rats. Biomed Res. 2019; 30(2):303-7 [DOI:10.35841/biomedicalresearch.30-19-063]
Kalra BS, Aggarwal S, Khurana N, Gupta U. Effect of cimetidine on hepatotoxicity induced by isoniazid-rifampicin combination in rabbits Bhupinder. Indian J Gastroenterol. 2007; 26(1):18-21. [PMID]
Peterson FJ, Knodell RG, Lindemann NJ, Steele NM. Prevention of acetaminophen and cocaine hepatotoxicity in mice by cimetidine treatment. Gastroenterology. 1983; 85(1):122-9. [DOI:10.1016/S0016-5085(83)80238-8] [PMID]
Jiang DW, Wang QR, Shen XR, He Y, Qian TT, Liu Q, et al. Radioprotective effects of cimetidine on rats irradiated by long-term, low-dose-rate neutrons and 60 Co γ-rays. Mil Med Res. 2017; 4:7. [DOI:10.1186/s40779-017-0116-7] [PMID] [PMCID]
Kaur G, Gan YL, Phillips CL, Wong K, Saini B. Chronotherapy in practice: The perspective of the community pharmacist. Int J Clin Pharm. 2016; 38(1):171-82. [DOI:10.1007/s11096-015-0228-7] [PMID]
Elliot HL, Meredith PA. Calculation of trough to peak ratio in a research unit setting advantages and disadvantages. Am J Hypertens. 1996; 9(10):71S-75S. [DOI:10.1016/S0895-7061(96)00266-X]
Ballesta A, Innominato PF, Dallmann R, Rand DA, Lévi FA. Systems chronotherapeutics. Pharmacol Rev. 2017; 69(2):161-99. [DOI:10.1124/pr.116.013441] [PMID] [PMCID]
Adikwu E, Braimbaifa N, Obianime AW. Melatonin and alpha lipoic acid: Possible mitigants for lopinavir/ritonavir- induced renal toxicity in male albino rats. Physiol Pharmacol. 2015; 19(4):232-40.
Nathiya S, Rajaram S, Abraham P. Hesperidin alleviates antitubercular drug induced oxidative stress, inflammation and apoptosis in rat liver. Int J Biomed Res. 2016; 7(7):439-46. [DOI:10.7439/ijbr.v7i7.3414]
The European Parliament And The Council Of The European :union:. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. Official Journal of the European :union:. 2010. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32010L0063
Al-Jawad FH, Abdulsahib WK, Al-Attar Z, Al-Hussaini JA. Protective effect of cimetidine, isosorbide dinitrate & vitamin c in experimental model of acute liver injury. Sch Acad J Pharm. 2018; 7(7):308-12. http://saspublisher.com/wp-content/uploads/2018/07/SAJP-77-308-312-c.pdf
Dalaklioglu S, Genc GE, Aksoy NH, Akcit F, Gumuslu S. Resveratrol ameliorates methotrexate-induced hepatotoxicity in rats via inhibition of lipid peroxidation. Hum Exp Toxicol. 2013; 32(6):662-71. [DOI:10.1177/0960327112468178] [PMID]
Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol. 1978; 52:302-10. [DOI:10.1016/S0076-6879(78)52032-6] [PMID]
Sun M, Zigma S. An improved spectrophotometer assay of superoxide dismutase based on epinephrine antioxidation. Anal Biochem. 1978; 90(1):81-9. [DOI:10.1016/0003-2697(78)90010-6] [PMID]
Aebi H. Catalase in vitro. Meth in Enzymol. 1984; 105:121- 6. [DOI:10.1016/S0076-6879(84)05016-3]
Rotruck JT, Rope AL, Ganther HF, Swason AB, Hafeman DG, Hoekstra WG. Selenium: Biochemical role as a component of glutathione peroxidase. Science. 1973; 179(4073):588-90. [DOI:10.1126/science.179.4073.588] [PMID]
Sedlak J, Lindsay RH. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem. 1968; 25:192-205. [DOI:10.1016/0003-2697(68)90092-4] [PMID]
Gornall AG, Bardawill CJ, David MM. Determination of serum proteins by means of the biureto reaction. J Biol Chem. 1949; 177:751-66. https://www.jbc.org/content/177/2/751.short
Bailey SA, Zidell RH, Perry RW. Relationships between organ weight and body/brainweight in the rat: What is the best analytical endpoint? Toxicol Pathol. 2004; 32(4):448-66.[DOI:10.1080/01926230490465874] [PMID]
Vroon DH, Israili Z. Aminotransferases. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The history, physical, and laboratory examinations. Boston: Butterworths; 1990. [PMID]
Elias SES, Eltom A, Osman AL, Babker AMA. Gamma glutamyl transferase and lactate dehydrogenase as biochemical markers of severity of preeclampsia among Sudanese pregnant women. Int J Reprod Contracept Obstet Gynecol. 2018; 7(8):3020-3. [DOI:10.18203/2320-1770.ijrcog20183294]
Hadi NR, Al-Amran FG, Swadi A. Metformin ameliorates methotrexate-induced hepatotoxicity. J Pharmacol Pharmacother. 2012; 3(3):248-53. [DOI:10.4103/0976-500X.99426] [PMID] [PMCID]
Tag HM. Hepatoprotective effect of mulberry (Morus nigra) leaves extract against methotrexate induced hepatotoxicity in male albino rat. BMC Comp and Altern Med. 2015; 15:252. [DOI:10.1186/s12906-015-0744-y] [PMID] [PMCID]
Halliwell B. Free radicals, antioxidants and human disease: Curiosity, cause or consequence? Lancet. 1994; 344(8924):721-4. [DOI:10.1016/S0140-6736(94)92211-X] [PMID]
Jahovic N, Cevik H, Sehirli OA, Yeğen BC, Sener G. Melatonin prevents methotrexate - induced hepatorenal oxidative injury in rats. J Pineal Res. 2003; 34(4):282-7. [DOI:10.1034/j.1600-079X.2003.00043.x] [PMID]
Gasparovic AC, Jaganjac M, Mihaljevic B, Sunjic SB, Zarkovic N. Assays for the measurement of lipid peroxidation.Methods Mol Biol. 2013; 965:283-96. [DOI:10.1007/978-1-62703-239-1_19] [PMID]
Kose E, Sapmaz HI, Sarihan E, Vardi N, Turkoz Y, Ekinci N. Beneficial effects of montelukast against methotrexate-induced liver toxicity: A biochemical and histological study. ScientificWorldJournal. 2012; 2012:987508. [DOI:10.1100/2012/987508] [PMID] [PMCID]
Soliman ME. Evaluation of the possible protective role of folic acid on the liver toxicity induced experimentally by methotrexate in adult male albino rats Egypt. J Histol. 2009; 1(1):118-28. https://www.sid.ir/en/journal/ViewPaper.aspx?ID=430087
Ros S, Juanola X, Condom E, Canas C, Riera J, Guardiola J, et al. Light and electron microscopic analysis of liver biopsy samples from rheumatoid arthritis patients receiving long-term methotrexate therapy. Scand J Rheumatol. 2002; 3(6):330-6. [DOI:10.1080/030097402320817040] [PMID]
Saka S, Aouacheri O. The investigation of the oxidative stressrelated parameters in high doses methotrexate-induced albino wistar rats. J Bioequiv Availab. 2017; 9(2):372-6. https://bit.ly/3dr7TYZ
Uchida K, Kawakishi S. Cimetidine anti-ulcer drug as a powerful hydroxyl radical scavenger. Agric and Biol Chem. 1990; 54(9):2485-7. [DOI:10.1271/bbb1961.54.2485]
Číž M, Lojek A. Modulation of neutrophil oxidative burst via histamine receptors. Br J Pharmacol. 2013; 170(1):17-22. [DOI:10.1111/bph.12107] [PMID] [PMCID]
Lambat Z, Limson JL, Daya S. Cimetidine: Antioxidant and metal‐binding properties. J Pharm Pharmacol. 2002; 54(12):1681-6. [DOI:10.1111/j.2042-7158.2002.tb02404.x] [PMID]
Shaik IH, Mehvar R. Effects of cytochrome p450 inhibition by cimetidine on the warm hepatic ischemia-reperfusion injury in rats. J Surg Res. 2010; 159(2):680-8. [DOI:10.1016/j.jss.2008.09.016] [PMID]
Furst DE, Kremer JM. Methotrexate in rheumatoid arthritis. Arthritis Rheum. 1988; 31(3):305-14. [DOI:10.1002/art.1780310301] [PMID]
Singh H, Pahwa S., Dhamija K, Arora V Cimetidine: A review. Int J Chem Tech Res. 2018; 11(02):115-23. [DOI:10.20902/IJCTR.2018.110944]
Type of Study: Original Research |
Subject:
Toxicology
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |