Volume 9, Issue 3 (2023)
Pharm Biomed Res 2023, 9(3): 173-182 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Suttithumsatid W, Kara J, Lomlim L, Nualsri C, Panichayupakaranant P. Acetylcholinesterase Inhibitory Activity of Standardized Cannabinoids-rich Fractions. Pharm Biomed Res 2023; 9 (3) :173-182
URL: http://pbr.mazums.ac.ir/article-1-454-en.html
URL: http://pbr.mazums.ac.ir/article-1-454-en.html
Wiwit Suttithumsatid1 

 , Jiraporn Kara2
, Jiraporn Kara2 

 , Luelak Lomlim2
, Luelak Lomlim2 

 , Charassri Nualsri3
, Charassri Nualsri3 

 , Pharkphoom Panichayupakaranant *1
, Pharkphoom Panichayupakaranant *1 




 , Jiraporn Kara2
, Jiraporn Kara2 

 , Luelak Lomlim2
, Luelak Lomlim2 

 , Charassri Nualsri3
, Charassri Nualsri3 

 , Pharkphoom Panichayupakaranant *1
, Pharkphoom Panichayupakaranant *1 


1- Department of Pharmacognosy and Pharmaceutical Botany, Faculty of Pharmaceutical Sciences, Prince of Songkla University, Hat-Yai, Thailand.
2- Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, Prince of Songkla University, Hat-Yai, Thailand.
3- Agricultural Innovation and Management Division, Faculty of Natural Resources, Prince of Songkla University, Hat-Yai, Thailand.
2- Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, Prince of Songkla University, Hat-Yai, Thailand.
3- Agricultural Innovation and Management Division, Faculty of Natural Resources, Prince of Songkla University, Hat-Yai, Thailand.
Full-Text [PDF 1815 kb]
(810 Downloads)
| Abstract (HTML) (2938 Views)
Full-Text: (738 Views)
Introduction
Alzheimer’s disease (AD), one of the most common forms of dementia, is a brain disorder that is manifested in disruption of thinking skills, memory impairment as well as disorder of language and visuospatial skills. The global prevalence of dementia is expected to increase gradually and may reach 96 million cases in 2050 [1]. In 2020, more than 5 million elderly Americans (>65 years old) had AD and this trend is projected to increase to nearly 14 million cases in the next 40 years. Death rates for AD are also increasing; recently, AD was reported as the sixth leading cause of death among US adults and the fifth leading cause of death among adults aged more than 65 years old [2-4].
The underlying pathophysiology of AD is an accumulation of abnormal neuritic plaques or β-amyloid plaques and neurofibrillary tangles that lead to the loss of function of cholinergic neurons in the brain and neurotransmission process. These processes trigger cognitive impairment in patients [5]. Acetylcholinesterase (AChE) is a cholinergic enzyme that is necessary for terminating neuronal transmission and signaling between synapses to restrict the activation of ACh with the specific receptor [6]. In AD patients, AChE level is increased around β-amyloid plaques; therefore, it may play an important role in β-amyloid fibrillogenesis. Importantly, the inhibition of AChE leads to the accumulation of acetylcholine. Elevated levels of AchE result in increased stimulation of muscarinic and nicotinic receptors, which leads to improvement in some clinical outcomes in AD [7, 8]. Recently, various AChE inhibitors have been discovered and are being used, such as donepezil, galantamine, and rivastigmine. However, there are some limitations in the use of these medications, especially with regard to efficacy as well as an adverse effect when used at a high dose [9].
After the recent announcement of cannabis legalization for medicinal purposes in many countries, medicinal cannabis has gained much attention, particularly in neurologic disorders. The most promising active compounds in Cannabis sativa L. are cannabinoids. The two major cannabinoids in C. sativa are CBD and THC. Both compounds have been reported to possess diverse pharmacological activities [10]. Cannabis extracts and/or cannabis oils with high content of cannabinoids are increasingly used as alternative treatments for several neurological disorders due to their interactions with the endocannabinoid system through many similar signaling pathways of neurodegenerative conditions [11]. However, the exact beneficial roles of pure cannabinoids as well as cannabis extract in AD patients are still emerging.
Although several conventional extraction methods, including maceration, percolation, heat reflux, and Soxhlet extraction, used for producing cannabis extracts or cannabis oils are available, these methods possess some disadvantages, such as the need for long extraction time and a large volume of solvent, which some of them are hazardous. Moreover, the heat-accelerated methods still need high temperatures during a long period of extraction leading to the loss of thermally labile compounds. Recently, Microwave-assisted extraction (MAE) has been popularized as a green extraction method for various phytochemicals due to its key advantages, such as less time, solvent, and energy consumption for extraction as well as high phytochemical content in herbal extracts [12, 13]. Therefore, the present study aimed to establish a green MAE method for the preparation of cannabinoid-rich extracts containing different concentrations of CBD and THC and evaluate their role in the modulation of the AChE inhibitory effect. The obtained finding may be relevant to the proper use of cannabinoid extracts in AD.
Materials and Methods
Chemicals and materials
Cannabidiol (CBD) was purchased from Chemfaces (Wuhan, China). Acetylcholine iodide, acetylcholinesterase enzyme from electric eel (Electrophorus electricus), 5,5´-dithiobis (2-nitrobenzoic acid), and Trizma® base were obtained from Sigma Aldrich (Singapore), while Silica gel 60 RP-18 F254s plate was from Merck Supelco Sigma-Aldrich (Mainz, Germany). Methanol (analytical and HPLC grades), ethanol, diethyl ether, chloroform, and hydrochloric acid were obtained from RCI Labscan (Bangkok, Thailand). Silica gel (60 A°, 60-200 µm) was purchased from Vertical Chromatography (Nonthaburi, Thailand), while Diaion® HP-20 was obtained from Mitsubishi Chemical Corporation (Tokyo, Japan). A Luna® C-18 column was purchased from Phenomenex (Bangkok, Thailand). A dried cannabis sample was obtained from the Narcotics Control Board, Bangkok, Thailand.
Preparation of cannabinoids-rich extract
Dried cannabis (Figure 1) was obtained from the Faculty of Natural Resources, Prince of Songkla University, and its voucher specimens (No. SKP 036 03 19 01) were kept at the herbarium of the Faculty of Pharmaceutical Sciences, Prince of Songkla University, Hat Yai, Thailand. Dried cannabis (920 g) was reduced to powder using a grinding machine (Retsch, Germany) and the powder was sieved through a No. 4 mesh to produce a moderately fine powder (780 g). The cannabis powder (750 g) was extracted with ethanol (1.5 L) using MAE (900 W) at 63°C for 9 minutet. After filtration, the solvent was evaporated using a rotary vacuum evaporator (Heidolph, Germany) to obtain a dark brown oily extract (106 g) from the filtrate.

The extract (10 g) was chromatographed on a 500 g Diaion® HP-20 column with gradient elution from 50% ethanol to 95% ethanol. Based on TLC analysis, the fractions containing CBD and THC were pooled to produce three cannabinoid-rich extracts, namely Fractions I, II, and III.
Purification of Δ-9-tetrahydrocannabinol
Δ-9-tetrahydrocannabinol (THC) was purified from the cannabinoid extract (2.6 g) using a silica gel column with a step-gradient elution of 2% and 10% diethyl ether in hexane, respectively. Fractions (20 mL) were collected. Based on thin-layer chromatographic (TLC) analysis, fractions 80-83 were the main fractions containing THC. They were pooled, the solvent was evaporated, and the sample was subsequently subjected to a silica gel column eluted with 40% dichloromethane in hexane. Fractions of 20 mL were collected. The pooled fractions 90-135 were dried and re-chromatographed on a silica gel column eluted with 10% diethyl ether in hexane. Fractions (20 mL) of eluent were collected. Pure THC (90 mg) was obtained from pooled fractions 10-11. The identity of THC was ascertained by 1H and 13C nuclear magnetic resonance (NMR) analysis at 500 MHz using the AscendTM NMR spectrometer (Bruker/Avance NEO, Switzerland).
TLC method
Standard compounds (CBD and THC) and samples were dissolved in methanol. A reversed-phase TLC technique was used for the detection of CBD and THC. The silica gel 60 RP-18 F254s plate was developed in methanol-water (85:15, v/v). Detection of CBD and THC was performed under UV 254 nm and sprayed with 10% sulfuric acid in methanol. The Rf values of CBD and THC were 0.44, and 0.27, respectively.
Quantitative HPLC method
Quantitative HPLC analysis of CBD and THC was performed using the method previously described by Saingam and Sakunpak [14], with some modifications. Separation was achieved isocratically on a reverse-phase Luna® C-18 column (4.6 mm×250 mm, 5 µm) equipped with a photodiode-array detector and autosampler (Shimadzu, Japan). The mobile phase consisted of methanol and water (85:15, v/v) and was eluted at a flow rate of 1 mL/min. HPLC chromatogram was determined using a UV–vis detector, at a wavelength of 220 nm. Limits of detection and quantification for THC were 0.12 and 0.40 µg/mL, and for CBD were 0.23 and 0.76 µg/mL, respectively.
Calibration curves of CBD and THC were established using authentic CBD and THC. A working solution of CBD and THC was diluted with methanol to produce six concentrations (6.25, 12.5, 25, 50, 100, and 200 µg/mL). The calibration curves were plotted between concentrations and peak area followed by linear regression analysis. In this study, the calibration curves of CBD and THC were Y=72615X+72146 (R2=0.9998) and Y=54467X+77267 (R2=0.9999), respectively.
Analyzed samples were prepared by accurately weighing the extracts (2.0 mg) and diluting them with methanol to 10 mL in a volumetric flask. The sample solutions were filtrated through a 0.45 µm filter prior to HPLC analysis.
Acetylcholinesterase inhibitory assay
Acetylcholinesterase inhibitory activity was evaluated using a photometric method modified from Ellman [15]. The incubation mixtures for the assay composed of 3 mM 5,5´-dithiobis (2-nitrobenzoic acid) (125 µL), 1.5 mM acetylcholine iodide (25 µL), 50 mM TRIS-HCl buffer pH 8.0 (50 µL), sample solution in ethanol (25 µL), and AChE enzyme (25 µL) in a 96-well plate. The absorbance of the solutions was measured at 405 nm every 11 seconds for 2 minutes using a PowerWaveX microplate reader (Biotek, USA). AChE inhibitory activity was analyzed using Prism Software and expressed as percentage inhibition and half maximal inhibitory concentration (IC50). Galantamine was used as a positive control. The experiment was performed in triplicate.
Statistical analysis
The results are expressed as Mean±SD. Statistical significance was calculated using a one-way analysis of variance (ANOVA) followed by Tukey’s multiple range test (P<0.05).
Results
Purification of THC
THC was purified from cannabis extract through three successive silica gel columns. The purity and identity of the purified compound were determined based on the HPLC method and revealed a single peak on the HPLC chromatogram, with a similar UV absorption spectrum (Figure 2) as the previous report for THC [16]. The chemical structure of the obtained compound was determined using 1H and 13C NMR data (Table 1) and compared with the previous report for THC [17].


Preparation of cannabinoids-rich extracts
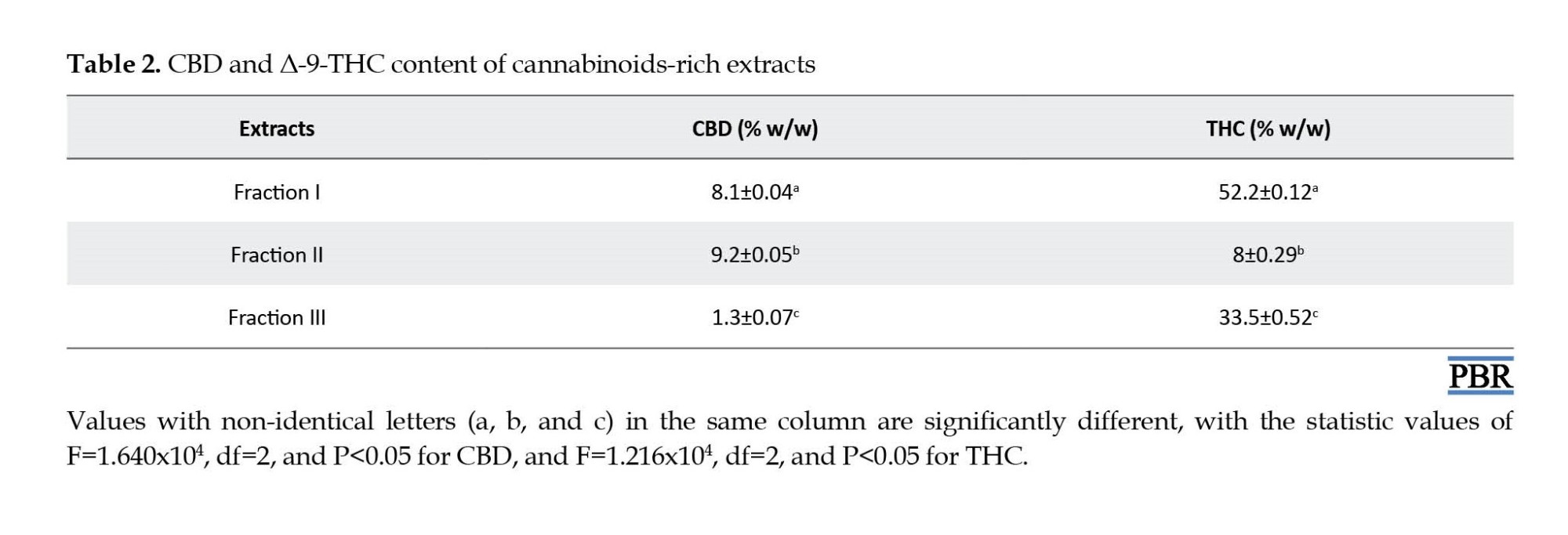
The cannabinoid-rich extracts were prepared using a green microwave extraction followed by a simple fractionation on a hydrophobic column. Threefractions with different concentrations of CBD and THC, namely Fraction I, II, and II were obtained. Based on HPLC analysis, CBD and THC were eluted at 5.0 and 10.1 min, respectively, with a high degree of resolution. The HPLC chromatograms of cannabinoids-rich extracts and standard compounds are shown in Figure 2 and their content of CBD and THC are presented in Table 2. Fraction I contained a moderate level of CBD (8.1% w/w) with a very high level of THC (52.2% w/w) with a CBD:THC ratio of about 1:6; Fraction II contained moderate levels of both CBD (9.2% w/w) and THC (8.0% w/w) at a CBD:THC ratio of about 1:1; and Fraction III contained a low level of CBD (1.3% w/w) and a high level of THC (33.5% w/w) with a CBD:THC ratio of about 1:26.
Acetylcholinesterase inhibitory activity
The results of in vitro anti-AChE activity of cannabinoid-rich fractions, CBD, and galantamine are shown in Table 3. Among the cannabinoids-rich extracts, Fraction I exhibited the highest activity followed by Fractions II and III with IC50 values of 52.3, 59.8, and 71.2 µg/mL, respectively. In addition, galantamine, a positive control, and CBD, a marker compound exhibited good AChE inhibitory activity with IC50 values of 0.7 and 13.7 µg/mL, respectively.

Discussion
Among neurologic disorders, AD has one of the highest incidences and is a top-ranking leading cause of death worldwide. Although there are numerous conventional medicines available for the treatment of AD, they come with some limitations, especially in terms of efficacy and safety. Therefore, more efficacious and safer remedies are still needed. Historically, natural products or phytochemical compounds have played an important role in the treatment of many human diseases. C. sativa extract and its two major cannabinoids, viz; CBD and THC, have been reported to possess diverse beneficial effects in the treatment of many diseases.
The resin extracts from C. sativa leaves reportedly demonstrate a dose-dependent inhibitory effect against AChE in both in vitro and in vivo studies. Interestingly, THC and CBD were expected to be potent AChE inhibitors based on in silico studies, with binding affinities of -10.3 and -9.8 kcal/mol, respectively. THC inhibited AChE via hydrogen bonding with Tyr120, pi-pi interaction with Trp286, and Van der Waals interaction with various amino acid residues, while CBD exhibited only Van der Waals interaction with various amino acid residues [18, 19]. However, no physiological interaction between THC and AChE was found in animal models [20]. Therefore, the present study focused on investigating the anti-AChE potential of cannabinoids-rich fractions containing different concentrations and ratios of CBD and THC, i.e. Fraction I that contained a moderate level of CBD with a very high level of THC at a CBD:THC ratio of about 1:6; Fraction II that contained the moderate levels of both CBD and THC at a ratio of about 1:1, and Fraction III contained a low level of CBD with a high level of THC at a ratio of about 1:26 and compared with CBD, a marker compound.
All three cannabinoid-rich extracts exhibited satisfactory anti-AChE activity, of which Fraction I exhibited the most potent activity, with an IC50 of 52.3 µg/mL. Fractions II with almost the same content of CBD but markedly lower content of THC than Fraction I exhibited slightly lower activity (IC50 of 59.8 µg/mL) than Fraction I. It seems to be the case that THC rarely contributes to the anti-AChE effect of cannabinoid-rich fractions. This was also confirmed by the anti-AChE activity of Fraction III (IC50 of 71.2 µg/mL), which is lower than Fraction II, even Fraction III contained a markedly higher content of THC. Moreover, CBD alone exhibited a good anti-AChE effect with IC50 13.8 µg/mL. This finding implied that CBD, but not THC plays a key role in the AChE inhibitory effect of the cannabis extracts. Other than the anti-AChE effect, CBD has been reported to activate the peroxisome proliferator-activated receptor-γ to protect damage from neurotoxicity and oxidation stress, suppress the process of abnormally hyperphosphorylation of tau protein and induce neurogenesis of the hippocampus area, which are beneficial in AD treatment [21, 22]. Additionally, when CBD was administered with THC into mice, it was reported that this combination treatment improved memory function better than treatment with each compound alone [22].
Regarding the relation between the content of CBD in the fractions (8.1-9.2% w/w) and their anti-AChE effect (IC50 of 52.3-59.8 µg/mL) compared with that of pure CBD (IC50 13.8 µg/mL), it seems that the fractions contained other active compounds. Because the extracts contained less than 10% w/w CBD but exhibited only 4-times less activity than CBD. Other cannabinoids, such as cannabinol (CBN), cannabicyclol, cannabitriol, cannabielosin, cannabugerol, cannabichromene, and cannabivarin have previously been recorded to display anti-AChE potential, in silico [19, 23]. In this study, the HPLC chromatograms of the cannabinoid-rich extracts also showed a peak at 8.2 minute (Figure 3), which could be identified as CBN, a cannabinoid commonly produced by the oxidation of THC. Indeed, the identity of the peak was confirmed by determining its UV absorption spectrum (Figure 4) produced by a photodiode array detector. When the spectrum was compared with that of the previous report for pure CBN, both were found to be similar with identical λmax [24]. Interestingly, although Fraction III contained a low content of CBD (1.3% w/w), its anti-AChE (IC50 of 71.2 µg/mL) was slightly lower than those of the others. This may be due to a high content of CBN in Fraction III as suggested by its HPLC chromatogram (Figure 3D).


The major pathologies of AD are presented by an inflammatory response in the brain, which is the main cause of the exacerbating β-amyloid burden and neurofibrillary tangles, suggesting that these two mechanisms correlate with neurologic disorder and cognitive decline in AD patients [5, 25]. The anti-AChE effect of cannabinoids possibly leads to an increased accumulation of acetylcholine and enhanced stimulation of cholinergic receptors, which play a crucial role in the cholinergic anti-inflammatory process. In addition, AChE inhibitors may suppress inflammation of the nervous system by diminishing the production of pro-inflammatory cytokines, such as TNF and IL-6 through the alpha7 nicotinic acetylcholine receptors [26-28]. Therefore, cannabinoids conceivably become an interesting candidate for alleviating the symptoms of AD.
Considering the efficacy of cannabis in AD therapy from pre-clinical studies, the use of cannabinoids, especially when CBD and THC are combined, might have greater benefits over a single pure cannabinoid compound in terms of improving dementia-related symptoms. The positive synergistic effect between cannabinoids as well as other compounds in cannabis, i.e. entourage effect, may play a crucial role in the use of cannabinoids-rich extracts with high content of many phytochemicals, including CBD in the alleviation of AChE-related challenges in AD.
Conclusion
In this cell-free study, we established that CBD as one of the major bioactive compounds in C. sativa is the leading agent regarding the inhibition of AChE activity. Although a previous in silico study predicted THC to have a high potential as an anti-AChE agent, our data indicated that THC only plays an adjunctive role, with CBD being the most important AChE inhibitory cannabinoid. Cannabinoids-rich extracts that contain at least 8% w/w CBD with low content THC might be considered as an alternative AChE inhibitor. However, further studies on the AChE inhibitory activity of the cannabinoids as well as other bioactive compounds in cannabis are needed to clearly identify the exact role of each component on the AChE inhibitory effect. Future high-quality research on animal models and clinical trials will also be needed to investigate the clinical efficacy and safety profile prior to any practical application of cannabinoid-rich extracts in AD patients.
Ethical Considerations
Compliance with ethical guidelines
There were no ethical considerations to be considered in this research.
Funding
This research was financially supported by PSU-PhD scholarship funded by Graduate School, Prince of Songkla University.
Authors' contributions
Conceptualization, data analysis, finding discussion and manuscript preparation: Wiwit Suttithumsatid, Pharkphoom Panichayupakaranan; Providing cannabis raw materials: Charassri Nualsri; Conducting experiments: Wiwit Suttithumsatid; Designing and carry out the acetylcholinesterase inhibitory assay: Jiraporn Kara and Luelak Lomlim.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors wish to thank Fredrick Eze for assistance with English editing.
References
Alzheimer’s disease (AD), one of the most common forms of dementia, is a brain disorder that is manifested in disruption of thinking skills, memory impairment as well as disorder of language and visuospatial skills. The global prevalence of dementia is expected to increase gradually and may reach 96 million cases in 2050 [1]. In 2020, more than 5 million elderly Americans (>65 years old) had AD and this trend is projected to increase to nearly 14 million cases in the next 40 years. Death rates for AD are also increasing; recently, AD was reported as the sixth leading cause of death among US adults and the fifth leading cause of death among adults aged more than 65 years old [2-4].
The underlying pathophysiology of AD is an accumulation of abnormal neuritic plaques or β-amyloid plaques and neurofibrillary tangles that lead to the loss of function of cholinergic neurons in the brain and neurotransmission process. These processes trigger cognitive impairment in patients [5]. Acetylcholinesterase (AChE) is a cholinergic enzyme that is necessary for terminating neuronal transmission and signaling between synapses to restrict the activation of ACh with the specific receptor [6]. In AD patients, AChE level is increased around β-amyloid plaques; therefore, it may play an important role in β-amyloid fibrillogenesis. Importantly, the inhibition of AChE leads to the accumulation of acetylcholine. Elevated levels of AchE result in increased stimulation of muscarinic and nicotinic receptors, which leads to improvement in some clinical outcomes in AD [7, 8]. Recently, various AChE inhibitors have been discovered and are being used, such as donepezil, galantamine, and rivastigmine. However, there are some limitations in the use of these medications, especially with regard to efficacy as well as an adverse effect when used at a high dose [9].
After the recent announcement of cannabis legalization for medicinal purposes in many countries, medicinal cannabis has gained much attention, particularly in neurologic disorders. The most promising active compounds in Cannabis sativa L. are cannabinoids. The two major cannabinoids in C. sativa are CBD and THC. Both compounds have been reported to possess diverse pharmacological activities [10]. Cannabis extracts and/or cannabis oils with high content of cannabinoids are increasingly used as alternative treatments for several neurological disorders due to their interactions with the endocannabinoid system through many similar signaling pathways of neurodegenerative conditions [11]. However, the exact beneficial roles of pure cannabinoids as well as cannabis extract in AD patients are still emerging.
Although several conventional extraction methods, including maceration, percolation, heat reflux, and Soxhlet extraction, used for producing cannabis extracts or cannabis oils are available, these methods possess some disadvantages, such as the need for long extraction time and a large volume of solvent, which some of them are hazardous. Moreover, the heat-accelerated methods still need high temperatures during a long period of extraction leading to the loss of thermally labile compounds. Recently, Microwave-assisted extraction (MAE) has been popularized as a green extraction method for various phytochemicals due to its key advantages, such as less time, solvent, and energy consumption for extraction as well as high phytochemical content in herbal extracts [12, 13]. Therefore, the present study aimed to establish a green MAE method for the preparation of cannabinoid-rich extracts containing different concentrations of CBD and THC and evaluate their role in the modulation of the AChE inhibitory effect. The obtained finding may be relevant to the proper use of cannabinoid extracts in AD.
Materials and Methods
Chemicals and materials
Cannabidiol (CBD) was purchased from Chemfaces (Wuhan, China). Acetylcholine iodide, acetylcholinesterase enzyme from electric eel (Electrophorus electricus), 5,5´-dithiobis (2-nitrobenzoic acid), and Trizma® base were obtained from Sigma Aldrich (Singapore), while Silica gel 60 RP-18 F254s plate was from Merck Supelco Sigma-Aldrich (Mainz, Germany). Methanol (analytical and HPLC grades), ethanol, diethyl ether, chloroform, and hydrochloric acid were obtained from RCI Labscan (Bangkok, Thailand). Silica gel (60 A°, 60-200 µm) was purchased from Vertical Chromatography (Nonthaburi, Thailand), while Diaion® HP-20 was obtained from Mitsubishi Chemical Corporation (Tokyo, Japan). A Luna® C-18 column was purchased from Phenomenex (Bangkok, Thailand). A dried cannabis sample was obtained from the Narcotics Control Board, Bangkok, Thailand.
Preparation of cannabinoids-rich extract
Dried cannabis (Figure 1) was obtained from the Faculty of Natural Resources, Prince of Songkla University, and its voucher specimens (No. SKP 036 03 19 01) were kept at the herbarium of the Faculty of Pharmaceutical Sciences, Prince of Songkla University, Hat Yai, Thailand. Dried cannabis (920 g) was reduced to powder using a grinding machine (Retsch, Germany) and the powder was sieved through a No. 4 mesh to produce a moderately fine powder (780 g). The cannabis powder (750 g) was extracted with ethanol (1.5 L) using MAE (900 W) at 63°C for 9 minutet. After filtration, the solvent was evaporated using a rotary vacuum evaporator (Heidolph, Germany) to obtain a dark brown oily extract (106 g) from the filtrate.

The extract (10 g) was chromatographed on a 500 g Diaion® HP-20 column with gradient elution from 50% ethanol to 95% ethanol. Based on TLC analysis, the fractions containing CBD and THC were pooled to produce three cannabinoid-rich extracts, namely Fractions I, II, and III.
Purification of Δ-9-tetrahydrocannabinol
Δ-9-tetrahydrocannabinol (THC) was purified from the cannabinoid extract (2.6 g) using a silica gel column with a step-gradient elution of 2% and 10% diethyl ether in hexane, respectively. Fractions (20 mL) were collected. Based on thin-layer chromatographic (TLC) analysis, fractions 80-83 were the main fractions containing THC. They were pooled, the solvent was evaporated, and the sample was subsequently subjected to a silica gel column eluted with 40% dichloromethane in hexane. Fractions of 20 mL were collected. The pooled fractions 90-135 were dried and re-chromatographed on a silica gel column eluted with 10% diethyl ether in hexane. Fractions (20 mL) of eluent were collected. Pure THC (90 mg) was obtained from pooled fractions 10-11. The identity of THC was ascertained by 1H and 13C nuclear magnetic resonance (NMR) analysis at 500 MHz using the AscendTM NMR spectrometer (Bruker/Avance NEO, Switzerland).
TLC method
Standard compounds (CBD and THC) and samples were dissolved in methanol. A reversed-phase TLC technique was used for the detection of CBD and THC. The silica gel 60 RP-18 F254s plate was developed in methanol-water (85:15, v/v). Detection of CBD and THC was performed under UV 254 nm and sprayed with 10% sulfuric acid in methanol. The Rf values of CBD and THC were 0.44, and 0.27, respectively.
Quantitative HPLC method
Quantitative HPLC analysis of CBD and THC was performed using the method previously described by Saingam and Sakunpak [14], with some modifications. Separation was achieved isocratically on a reverse-phase Luna® C-18 column (4.6 mm×250 mm, 5 µm) equipped with a photodiode-array detector and autosampler (Shimadzu, Japan). The mobile phase consisted of methanol and water (85:15, v/v) and was eluted at a flow rate of 1 mL/min. HPLC chromatogram was determined using a UV–vis detector, at a wavelength of 220 nm. Limits of detection and quantification for THC were 0.12 and 0.40 µg/mL, and for CBD were 0.23 and 0.76 µg/mL, respectively.
Calibration curves of CBD and THC were established using authentic CBD and THC. A working solution of CBD and THC was diluted with methanol to produce six concentrations (6.25, 12.5, 25, 50, 100, and 200 µg/mL). The calibration curves were plotted between concentrations and peak area followed by linear regression analysis. In this study, the calibration curves of CBD and THC were Y=72615X+72146 (R2=0.9998) and Y=54467X+77267 (R2=0.9999), respectively.
Analyzed samples were prepared by accurately weighing the extracts (2.0 mg) and diluting them with methanol to 10 mL in a volumetric flask. The sample solutions were filtrated through a 0.45 µm filter prior to HPLC analysis.
Acetylcholinesterase inhibitory assay
Acetylcholinesterase inhibitory activity was evaluated using a photometric method modified from Ellman [15]. The incubation mixtures for the assay composed of 3 mM 5,5´-dithiobis (2-nitrobenzoic acid) (125 µL), 1.5 mM acetylcholine iodide (25 µL), 50 mM TRIS-HCl buffer pH 8.0 (50 µL), sample solution in ethanol (25 µL), and AChE enzyme (25 µL) in a 96-well plate. The absorbance of the solutions was measured at 405 nm every 11 seconds for 2 minutes using a PowerWaveX microplate reader (Biotek, USA). AChE inhibitory activity was analyzed using Prism Software and expressed as percentage inhibition and half maximal inhibitory concentration (IC50). Galantamine was used as a positive control. The experiment was performed in triplicate.
Statistical analysis
The results are expressed as Mean±SD. Statistical significance was calculated using a one-way analysis of variance (ANOVA) followed by Tukey’s multiple range test (P<0.05).
Results
Purification of THC
THC was purified from cannabis extract through three successive silica gel columns. The purity and identity of the purified compound were determined based on the HPLC method and revealed a single peak on the HPLC chromatogram, with a similar UV absorption spectrum (Figure 2) as the previous report for THC [16]. The chemical structure of the obtained compound was determined using 1H and 13C NMR data (Table 1) and compared with the previous report for THC [17].


Preparation of cannabinoids-rich extracts
The cannabinoid-rich extracts were prepared using a green microwave extraction followed by a simple fractionation on a hydrophobic column. Threefractions with different concentrations of CBD and THC, namely Fraction I, II, and II were obtained. Based on HPLC analysis, CBD and THC were eluted at 5.0 and 10.1 min, respectively, with a high degree of resolution. The HPLC chromatograms of cannabinoids-rich extracts and standard compounds are shown in Figure 2 and their content of CBD and THC are presented in Table 2. Fraction I contained a moderate level of CBD (8.1% w/w) with a very high level of THC (52.2% w/w) with a CBD:THC ratio of about 1:6; Fraction II contained moderate levels of both CBD (9.2% w/w) and THC (8.0% w/w) at a CBD:THC ratio of about 1:1; and Fraction III contained a low level of CBD (1.3% w/w) and a high level of THC (33.5% w/w) with a CBD:THC ratio of about 1:26.

Acetylcholinesterase inhibitory activity
The results of in vitro anti-AChE activity of cannabinoid-rich fractions, CBD, and galantamine are shown in Table 3. Among the cannabinoids-rich extracts, Fraction I exhibited the highest activity followed by Fractions II and III with IC50 values of 52.3, 59.8, and 71.2 µg/mL, respectively. In addition, galantamine, a positive control, and CBD, a marker compound exhibited good AChE inhibitory activity with IC50 values of 0.7 and 13.7 µg/mL, respectively.

Discussion
Among neurologic disorders, AD has one of the highest incidences and is a top-ranking leading cause of death worldwide. Although there are numerous conventional medicines available for the treatment of AD, they come with some limitations, especially in terms of efficacy and safety. Therefore, more efficacious and safer remedies are still needed. Historically, natural products or phytochemical compounds have played an important role in the treatment of many human diseases. C. sativa extract and its two major cannabinoids, viz; CBD and THC, have been reported to possess diverse beneficial effects in the treatment of many diseases.
The resin extracts from C. sativa leaves reportedly demonstrate a dose-dependent inhibitory effect against AChE in both in vitro and in vivo studies. Interestingly, THC and CBD were expected to be potent AChE inhibitors based on in silico studies, with binding affinities of -10.3 and -9.8 kcal/mol, respectively. THC inhibited AChE via hydrogen bonding with Tyr120, pi-pi interaction with Trp286, and Van der Waals interaction with various amino acid residues, while CBD exhibited only Van der Waals interaction with various amino acid residues [18, 19]. However, no physiological interaction between THC and AChE was found in animal models [20]. Therefore, the present study focused on investigating the anti-AChE potential of cannabinoids-rich fractions containing different concentrations and ratios of CBD and THC, i.e. Fraction I that contained a moderate level of CBD with a very high level of THC at a CBD:THC ratio of about 1:6; Fraction II that contained the moderate levels of both CBD and THC at a ratio of about 1:1, and Fraction III contained a low level of CBD with a high level of THC at a ratio of about 1:26 and compared with CBD, a marker compound.
All three cannabinoid-rich extracts exhibited satisfactory anti-AChE activity, of which Fraction I exhibited the most potent activity, with an IC50 of 52.3 µg/mL. Fractions II with almost the same content of CBD but markedly lower content of THC than Fraction I exhibited slightly lower activity (IC50 of 59.8 µg/mL) than Fraction I. It seems to be the case that THC rarely contributes to the anti-AChE effect of cannabinoid-rich fractions. This was also confirmed by the anti-AChE activity of Fraction III (IC50 of 71.2 µg/mL), which is lower than Fraction II, even Fraction III contained a markedly higher content of THC. Moreover, CBD alone exhibited a good anti-AChE effect with IC50 13.8 µg/mL. This finding implied that CBD, but not THC plays a key role in the AChE inhibitory effect of the cannabis extracts. Other than the anti-AChE effect, CBD has been reported to activate the peroxisome proliferator-activated receptor-γ to protect damage from neurotoxicity and oxidation stress, suppress the process of abnormally hyperphosphorylation of tau protein and induce neurogenesis of the hippocampus area, which are beneficial in AD treatment [21, 22]. Additionally, when CBD was administered with THC into mice, it was reported that this combination treatment improved memory function better than treatment with each compound alone [22].
Regarding the relation between the content of CBD in the fractions (8.1-9.2% w/w) and their anti-AChE effect (IC50 of 52.3-59.8 µg/mL) compared with that of pure CBD (IC50 13.8 µg/mL), it seems that the fractions contained other active compounds. Because the extracts contained less than 10% w/w CBD but exhibited only 4-times less activity than CBD. Other cannabinoids, such as cannabinol (CBN), cannabicyclol, cannabitriol, cannabielosin, cannabugerol, cannabichromene, and cannabivarin have previously been recorded to display anti-AChE potential, in silico [19, 23]. In this study, the HPLC chromatograms of the cannabinoid-rich extracts also showed a peak at 8.2 minute (Figure 3), which could be identified as CBN, a cannabinoid commonly produced by the oxidation of THC. Indeed, the identity of the peak was confirmed by determining its UV absorption spectrum (Figure 4) produced by a photodiode array detector. When the spectrum was compared with that of the previous report for pure CBN, both were found to be similar with identical λmax [24]. Interestingly, although Fraction III contained a low content of CBD (1.3% w/w), its anti-AChE (IC50 of 71.2 µg/mL) was slightly lower than those of the others. This may be due to a high content of CBN in Fraction III as suggested by its HPLC chromatogram (Figure 3D).


The major pathologies of AD are presented by an inflammatory response in the brain, which is the main cause of the exacerbating β-amyloid burden and neurofibrillary tangles, suggesting that these two mechanisms correlate with neurologic disorder and cognitive decline in AD patients [5, 25]. The anti-AChE effect of cannabinoids possibly leads to an increased accumulation of acetylcholine and enhanced stimulation of cholinergic receptors, which play a crucial role in the cholinergic anti-inflammatory process. In addition, AChE inhibitors may suppress inflammation of the nervous system by diminishing the production of pro-inflammatory cytokines, such as TNF and IL-6 through the alpha7 nicotinic acetylcholine receptors [26-28]. Therefore, cannabinoids conceivably become an interesting candidate for alleviating the symptoms of AD.
Considering the efficacy of cannabis in AD therapy from pre-clinical studies, the use of cannabinoids, especially when CBD and THC are combined, might have greater benefits over a single pure cannabinoid compound in terms of improving dementia-related symptoms. The positive synergistic effect between cannabinoids as well as other compounds in cannabis, i.e. entourage effect, may play a crucial role in the use of cannabinoids-rich extracts with high content of many phytochemicals, including CBD in the alleviation of AChE-related challenges in AD.
Conclusion
In this cell-free study, we established that CBD as one of the major bioactive compounds in C. sativa is the leading agent regarding the inhibition of AChE activity. Although a previous in silico study predicted THC to have a high potential as an anti-AChE agent, our data indicated that THC only plays an adjunctive role, with CBD being the most important AChE inhibitory cannabinoid. Cannabinoids-rich extracts that contain at least 8% w/w CBD with low content THC might be considered as an alternative AChE inhibitor. However, further studies on the AChE inhibitory activity of the cannabinoids as well as other bioactive compounds in cannabis are needed to clearly identify the exact role of each component on the AChE inhibitory effect. Future high-quality research on animal models and clinical trials will also be needed to investigate the clinical efficacy and safety profile prior to any practical application of cannabinoid-rich extracts in AD patients.
Ethical Considerations
Compliance with ethical guidelines
There were no ethical considerations to be considered in this research.
Funding
This research was financially supported by PSU-PhD scholarship funded by Graduate School, Prince of Songkla University.
Authors' contributions
Conceptualization, data analysis, finding discussion and manuscript preparation: Wiwit Suttithumsatid, Pharkphoom Panichayupakaranan; Providing cannabis raw materials: Charassri Nualsri; Conducting experiments: Wiwit Suttithumsatid; Designing and carry out the acetylcholinesterase inhibitory assay: Jiraporn Kara and Luelak Lomlim.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors wish to thank Fredrick Eze for assistance with English editing.
References
- Reitz C, Mayeux R. Alzheimer disease: Epidemiology, diagnostic criteria, risk factors and biomarkers. Biochem Pharmacol. 2014; 88(4):640-51. [DOI:10.1016/j.bcp.2013.12.024] [PMID] [PMCID]
- Matthews KA, Xu W, Gaglioti AH, Holt JB, Croft JB, Mack D, et al. Racial and ethnic estimates of alzheimer’s disease and related dementias in the United States (2015-2060) in adults aged≥65 years. Alzheimers Dement. 2019; 15:17-24. [DOI:10.1016/j.jalz.2018.06.3063] [PMID] [PMCID]
- Xu J, Kochanek KD, Murphy SL, Tejada-Vera B. Deaths: Final data for 2007. Natl Vital Stat Rep. 2010; 58(19):1-19. [PMID]
- Heron MP. Deaths: Leading causes for 2017. Natl Vital Stat Rep. 2019; 68(6):1-77. [PMID]
- DeTure MA, Dickson DW. The neuropathological diagnosis of alzheimer’s disease. Mol Neurodegener. 2019; 14(1):32. [DOI:10.1186/s13024-019-0333-5] [PMID] [PMCID]
- McHardy SF, Wang H-YL, McCowen SV, Valdez MC. Recent advances in acetylcholinesterase inhibitors and reactivators: An update on the patent literature (2012-2015). Expert Opin Ther Pat. 2017; 27:455-76. [DOI:10.1080/13543776.2017.1272571] [PMID]
- Carvajal FJ, Inestrosa NC. Interactions of AChE with Aβ aggregates in alzheimer’s brain: Therapeutic relevance of IDN 5706. Front Mol Neurosci. 2011; 4:19. [DOI:10.3389/fnmol.2011.00019] [PMID] [PMCID]
- Lazarevic-Pasti T, Leskovac A, Momic T, Petrovic S, Vasic V. Modulators of acetylcholinesterase activity: From alzheimer’s disease to anti-cancer drugs. Curr Med Chem. 2017; 24:3283-309. [DOI:10.2174/0929867324666170705123509] [PMID]
- Ruangritchankul S, Chantharit P, Srisuma S, Gray LC. Adverse drug reactions of acetylcholinesterase inhibitors in older people living with dementia: A comprehensive literature review. Ther Clin Risk Manag. 2021; 17:927-49. [DOI:10.2147/TCRM.S323387] [PMID] [PMCID]
- Maayah ZH, Takahara S, Ferdaoussi M, Dyck JR. The molecular mechanisms that underpin the biological benefits of full-spectrum cannabis extract in the treatment of neuropathic pain and inflammation. Biochim Biophys Acta Mol Basis Dis. 2020; 1866(7):165771. [DOI:10.1016/j.bbadis.2020.165771] [PMID]
- Maccarrone M, Bernardi G, Agrò AF, Centonze D. Cannabinoid receptor signalling in neurodegenerative diseases: A potential role for membrane fluidity disturbance. Br J Pharmacol. 2011; 163:1379-90. [DOI:10.1111/j.1476-5381.2011.01277.x] [PMID] [PMCID]
- Radoiu M, Kaur H, Bakowska-Barczak A, Splinter S. Microwave-assisted industrial scale cannabis extraction. Technologies. 2020; 8:45. [DOI:10.3390/technologies8030045]
- Chang CW, Yen CC, Wu MT, Hsu MC, Wu YT. Microwave-assisted extraction of cannabinoids in hemp nut using response surface methodology: Optimization and comparative study. Molecules 2017; 22(11):1894. [DOI:10.3390/molecules22111894] [PMID] [PMCID]
- Saingam W, Sakunpak A. Development and validation of reverse phase high performance liquid chromatography method for the determination of delta-9-tetrahydrocannabinol and cannabidiol in oromucosal spray from cannabis extract. Rev Bras Farmacogn. 2018; 28:669-72. [DOI:10.1016/j.bjp.2018.08.001]
- Ellman G, Courtney K, Andres J, Featherstone R. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961; 7:88-95. [DOI:10.1016/0006-2952(61)90145-9] [PMID]
- Vyas G, Bhatt S, Paul P. Synthesis of calixarene-capped silver nanoparticles for colorimetric and amperometric detection of mercury (Hg II, Hg 0). ACS Omega 2019; 4:3860-70. [DOI:10.1021/acsomega.8b03299] [PMID] [PMCID]
- Choi YH, Hazekamp A, Peltenburg‐Looman AM, Frédérich M, Erkelens C, Lefeber AW, et al. NMR assignments of the major cannabinoids and cannabi flavonoids isolated from flowers of Cannabis sativa. Phytochem Anal. 2004; 15:345-54. [DOI:10.1002/pca.787] [PMID]
- Karimi I, Yousofvand N, Hussein BA. In vitro cholinesterase inhibitory action of cannabis sativa l. Cannabaceae and in silico study of its selected phytocompounds. In Silico Pharmacol. 2021; 9:1-15. [DOI:10.1007/s40203-021-00075-0] [PMID] [PMCID]
- Furqan T, Batool S, Habib R, Shah M, Kalasz H, Darvas F, et al. Cannabis constituents and acetylcholinesterase interaction: Molecular docking, in vitro studies and association with CNR1 rs806368 and ACHE rs17228602. Biomolecules. 2020; 10(5):758. [DOI:10.3390/biom10050758] [PMID] [PMCID]
- Moss D, Peck P, Salome R. Tetrahydrocannabinol and acetylcholinesterase. Pharmacol Biochem Behav. 1978; 8:763-5. [DOI:10.1016/0091-3057(78)90280-0] [PMID]
- Esposito G, Scuderi C, Valenza M, Togna G, Latina V. Cannabidiol reduces Aβ-induced neuroinflammation and promotes hippocampal. Plos One. 2011; 6:e28668. [DOI:10.1371/journal.pone.0028668] [PMID] [PMCID]
- Kim SH, Yang JW, Kim KH, Kim JU, Yook TH. A review on studies of marijuana for alzheimer’s disease-focusing on CBD, THC. Pharmacopuncture. 2019; 22(4):225-30. [DOI:10.3831/KPI.2019.22.030] [PMID] [PMCID]
- Seniya C, Khan GJ, Uchadia K. Identification of potential herbal inhibitor of acetylcholinesterase associated alzheimer’s disorders using molecular docking and molecular dynamics simulation. Biochem Res Int 2014; 2014:705451. [DOI:10.1155/2014/705451] [PMID] [PMCID]
- Hazekamp A, Peltenburg A, Verpoorte R, Giroud C. Chromatographic and spectroscopic data of cannabinoids from cannabis sativa l. J Liq Chromatogr Relat. 2005; 28:2361-82. [DOI:10.1080/10826070500187558]
- Kinney JW, Bemiller SM, Murtishaw AS, Leisgang AM, Salazar AM, Lamb BT. Inflammation as a central mechanism in alzheimer’s disease. Alzheimers Dement. 2018; 4:575-90. [DOI:10.1016/j.trci.2018.06.014] [PMID] [PMCID]
- Kamkwalala AR, Newhouse PA. Beyond acetylcholinesterase inhibitors: Novel cholinergic treatments for alzheimer’s disease. Curr Alzheimer Res. 2017; 14:377-92. [DOI:10.2174/1567205013666160930112625] [PMID]
- Rosas-Ballina M, Tracey KJ. Cholinergic control of inflammation. J Intern Med. 2009; 265(6):663-79. [DOI:10.1111/j.1365-2796.2009.02098.x] [PMID] [PMCID]
- Seyedabadi M, Rahimian R, Ghia JE. The role of alpha7 nicotinic acetylcholine receptors in inflammatory bowel disease: Involvement of different cellular pathways. Expert Opin Ther Targets. 2018; 22(2):161-76. [DOI:10.1080/14728222.2018.1420166] [PMID]
Type of Study: Original Research |
Subject:
Phyochemistry
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |






